Choosing Between Sn2 Sn1 E2 And E1 Reactions

Sn1 Sn2 E1 And E2 Reactions In Oc Pdf Chemical Reactions Unit The main difference between sn1, sn2, e1, and e2 reactions lies in their reaction type (substitution vs elimination), molecularity (uni or bimolecular), mechanism steps (single or multiple), and reagent type (nucleophile vs base). For a certain substrate, it may have chance to go through any of the four reaction pathways. so it seems rather challenging to predict the outcome of a certain reaction. we will talk about the strategies that can be applied in solving such problem, and explain the reasonings behind.
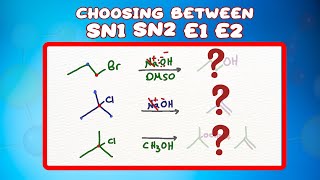
Choosing Between Sn2 Sn1 E2 And E1 Reactions Doovi An exam gives you a substrate, a nucleophile base, and a solvent, and you're supposed to predict whether the reaction runs sn1, sn2, e1, or e2. sometimes it's a mix. sometimes there's no reaction at all. there are four things to check, in order: substrate, nucleophile base, solvent, and temperature. In summary, follow these steps to identify if the mechanism is s n 1, s n 2, e1, or e2: 1) determine if the base nu is strong or weak if strong – sn2 or e2 if weak – sn1 or e1 2) if it is a strong, bulky base – e2 only. Look carefully in each reaction at the structure of the substrate, the leaving group, the nucleophile, and the solvent. then decide from the preceding summary which kind of reaction is likely to be favored. Before asking any other questions, look at the reactions below and see if you can identify potential substrates for s n 1 s n 2 e1 e2 reactions. let’s start with a fairly easy one.
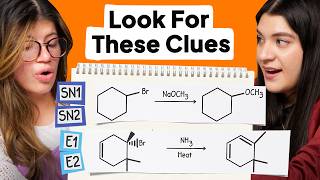
Choosing Between Sn2 Sn1 E2 And E1 Reactions Doovi Look carefully in each reaction at the structure of the substrate, the leaving group, the nucleophile, and the solvent. then decide from the preceding summary which kind of reaction is likely to be favored. Before asking any other questions, look at the reactions below and see if you can identify potential substrates for s n 1 s n 2 e1 e2 reactions. let’s start with a fairly easy one. If you haven’t seen my videos on the sn1, sn2, e1, and e2 mechanisms, i strongly encourage you to first watch those and then come back to this video. so, let’s start by a quick review of the important features of each mechanism. Master sn1, sn2, e1, and e2 reactions! learn to identify reactions by nucleophile, base strength, substrate, solvent, and predict products with examples. It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1 since both s n 2 and e2 require a strong nucleophile or strong base, which are usually negatively charged species, while s n 1 e1 requires neutral conditions. Sn2 , sn1 , e2 , & e1: substitution and elimination reactions nucleophilic substitution reactions (sn2 and sn1) replace a leaving group with a nucleophile (nu: or nu: ) elimination reactions (e2 and e1) generate a double bond by loss of " a " and " b: " they may compete with each other.

Difference Between Sn1 And Sn2 Reactions If you haven’t seen my videos on the sn1, sn2, e1, and e2 mechanisms, i strongly encourage you to first watch those and then come back to this video. so, let’s start by a quick review of the important features of each mechanism. Master sn1, sn2, e1, and e2 reactions! learn to identify reactions by nucleophile, base strength, substrate, solvent, and predict products with examples. It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1 since both s n 2 and e2 require a strong nucleophile or strong base, which are usually negatively charged species, while s n 1 e1 requires neutral conditions. Sn2 , sn1 , e2 , & e1: substitution and elimination reactions nucleophilic substitution reactions (sn2 and sn1) replace a leaving group with a nucleophile (nu: or nu: ) elimination reactions (e2 and e1) generate a double bond by loss of " a " and " b: " they may compete with each other.

Difference Between Sn1 And Sn2 Reactions It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1 since both s n 2 and e2 require a strong nucleophile or strong base, which are usually negatively charged species, while s n 1 e1 requires neutral conditions. Sn2 , sn1 , e2 , & e1: substitution and elimination reactions nucleophilic substitution reactions (sn2 and sn1) replace a leaving group with a nucleophile (nu: or nu: ) elimination reactions (e2 and e1) generate a double bond by loss of " a " and " b: " they may compete with each other.
Comments are closed.