Chemistry Separation Methods Simple And Fractional Distillation

Chemistry Separation Methods Simple And Fractional Distillation For igcse chemistry, learn about separation techniques like simple and fractional distillation, filtration, crystallisation and chromatography, with examples. Two common types of distillation methods are fractional distillation and simple distillation. while both techniques aim to achieve separation, they differ in terms of efficiency, complexity, and the types of mixtures they are best suited for.

Chemistry Water Distillation Process Fractional Distillation Simple There are different ways to separate mixtures, such as filtration, crystallisation, simple distillation, fractional distillation and chromatography. the method chosen depends on the type. Learn about simple and fractional distillation techniques. understand how to separate liquids with different boiling points and liquids from solutions. explore the apparatus, process, and key points for gcse chemistry and science. Learn about the technique of distillation, how and why it works, and its applications in laboratory settings and industry environments!. When it comes to separating multiple solvents or purifying a mixture, two common distillation methods are often employed: simple distillation and fractional distillation.
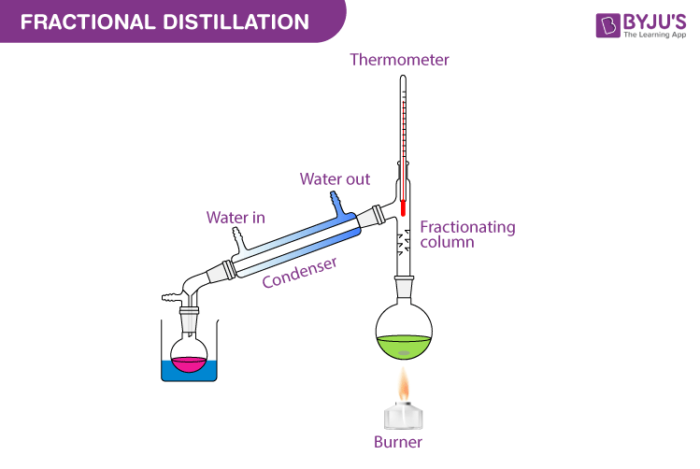
Fractional Distillation Detailed Explanation Along With Diagrams Learn about the technique of distillation, how and why it works, and its applications in laboratory settings and industry environments!. When it comes to separating multiple solvents or purifying a mixture, two common distillation methods are often employed: simple distillation and fractional distillation. The main difference between fractional distillation and simple distillation is that simple distillation separate liquids with boiling point gaps of at least 50 degrees whereas fractional distillation separates liquids with closer boiling points. See how these techniques are applied in the lab to prepare samples and in industry to produce fuels, foods, and medicines. with clear explanations, worked examples, and real world context, this resource builds both your knowledge and your practical skills for exams and beyond. However, the components of homogeneous mixtures cannot be separated by these simple separating techniques. for example, the air is a homogeneous mixture, and fractional distillation is the most common method to separate its components. Objective: in this experiment, students will analyze the efficiency of two separation techniques: simple and fractional distillation. plotting the temperature of distillate against the volume collected will allow students to compare the efficiency of both methods.
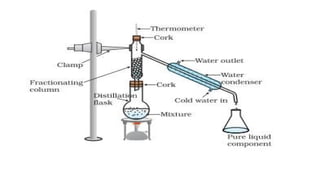
Simple Vs Fractional Distillation Methods Of Separation Pptx The main difference between fractional distillation and simple distillation is that simple distillation separate liquids with boiling point gaps of at least 50 degrees whereas fractional distillation separates liquids with closer boiling points. See how these techniques are applied in the lab to prepare samples and in industry to produce fuels, foods, and medicines. with clear explanations, worked examples, and real world context, this resource builds both your knowledge and your practical skills for exams and beyond. However, the components of homogeneous mixtures cannot be separated by these simple separating techniques. for example, the air is a homogeneous mixture, and fractional distillation is the most common method to separate its components. Objective: in this experiment, students will analyze the efficiency of two separation techniques: simple and fractional distillation. plotting the temperature of distillate against the volume collected will allow students to compare the efficiency of both methods.

Simple Vs Fractional Distillation Methods Of Separation Pptx However, the components of homogeneous mixtures cannot be separated by these simple separating techniques. for example, the air is a homogeneous mixture, and fractional distillation is the most common method to separate its components. Objective: in this experiment, students will analyze the efficiency of two separation techniques: simple and fractional distillation. plotting the temperature of distillate against the volume collected will allow students to compare the efficiency of both methods.
Comments are closed.