Chemistry Liquids And Solids 23 Of 59 Structures Properties Of H2o Water Dissolves Co2

Chemistry Of Water Pdf Water Fluid Dynamics Visit ilectureonline for more math and science lectures! in this video i will explain the structures and properties of water (water dissolves co2) .more. Conclusion: water's ability to dissolve carbon dioxide and regulate its levels in the atmosphere demonstrates another remarkable property of water and its significance in the earth's environmental processes.

Properties Of Solids Liquids And Gases Class 9 Science In 2024 Lecture 24: structures & properties of h2o: where does it come from?. We review how these properties are encoded within water’s molecular structure and energies, as understood from theories, simulations, and experiments. like simpler liquids, water molecules are nearly spherical and interact with each other through van der waals forces. Water is liquid at room temperature so it's able to move around quicker than it is as solid, enabling the molecules to form fewer hydrogen bonds resulting in the molecules being packed more closely together. That property is critical to life on earth as we think of how water based (aqueous) solutions support life in the seas and on land. we will discuss the energy changes that allow substances that accompany solution formation, and the effect of temperature and pressure on dissolution.

Liquids And Solids Ppt Water is liquid at room temperature so it's able to move around quicker than it is as solid, enabling the molecules to form fewer hydrogen bonds resulting in the molecules being packed more closely together. That property is critical to life on earth as we think of how water based (aqueous) solutions support life in the seas and on land. we will discuss the energy changes that allow substances that accompany solution formation, and the effect of temperature and pressure on dissolution. Chemistry liquids and solids (24 of 59) structures & properties of h2o: where does it come from?. Chemistry liquids and solids (24 of 59) structures & properties of h2o: where does it come from?. It begins by stating the learning objectives which are to explain water's properties based on its molecular structure and intermolecular forces, and describe the differences between crystalline and amorphous solids. Conclusion: water's high melting and boiling points, driven by its molecular structure and hydrogen bonding, are among the many remarkable properties that make it indispensable for life.
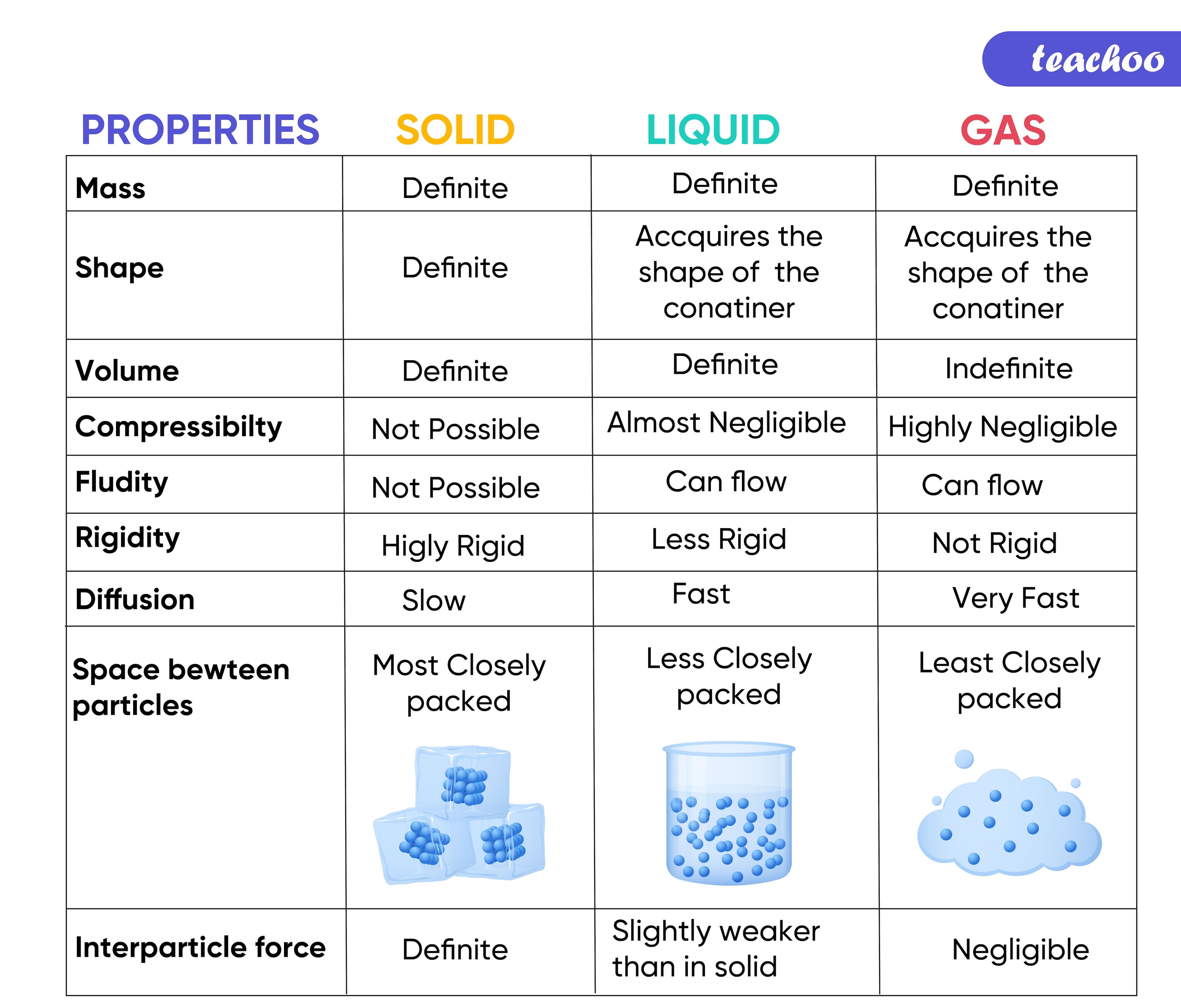
Properties Of Solids Liquids Gases Compared Teachoo Science Gasrebatenet Chemistry liquids and solids (24 of 59) structures & properties of h2o: where does it come from?. Chemistry liquids and solids (24 of 59) structures & properties of h2o: where does it come from?. It begins by stating the learning objectives which are to explain water's properties based on its molecular structure and intermolecular forces, and describe the differences between crystalline and amorphous solids. Conclusion: water's high melting and boiling points, driven by its molecular structure and hydrogen bonding, are among the many remarkable properties that make it indispensable for life.

Properties Of Solids Liquids Gases Compared Teachoo Science Gasrebatenet It begins by stating the learning objectives which are to explain water's properties based on its molecular structure and intermolecular forces, and describe the differences between crystalline and amorphous solids. Conclusion: water's high melting and boiling points, driven by its molecular structure and hydrogen bonding, are among the many remarkable properties that make it indispensable for life.
Comments are closed.