Chemical Reactions 3 Of 11 Combustion Reactions An Explanation

Chemical Reactions 3 Of 11 Combustion Reactions An Explanation A combustion reaction is a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat. combustion reactions must involve o a 2 as one reactant. Describes the basics of combustion reactions, how to identify them, predict the products and balance the chemical equation. three explosions are included, me.
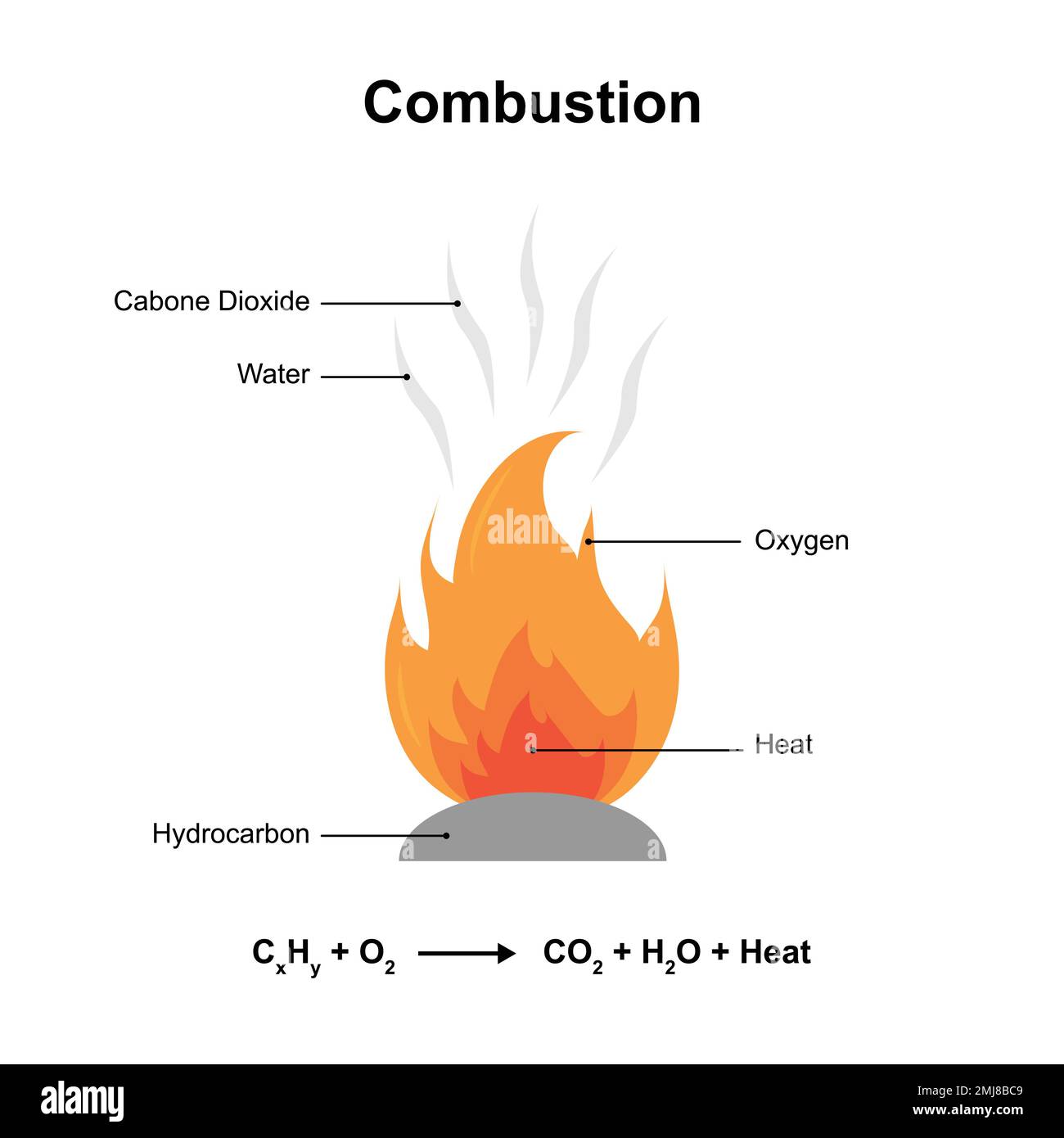
Types Of Chemical Reactions Combustion Displacement 40 Off Discover what combustion reactions are, how fuel reacts with oxygen to produce oxides, heat, and light, with key examples like magnesium, sulfur, methane, and propane explained clearly. Combustion, with rare exceptions, is a complex chemical process involving many steps that depend on the properties of the combustible substance. it is initiated by external factors such as heat, light, and sparks. the reaction sets in as the mixture of combustibles attains the ignition temperature. A combustion reaction is a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat. combustion reactions must involve o 2 as one reactant. What is a combustion reaction? a combustion reaction is an exothermic chemical reaction between substances, usually including oxygen gas and accompanied by the generation of heat, energy, and light (flame). the products of a combustion reaction depend on the combusted substance.

Combustion Reactions Ppt A combustion reaction is a reaction in which a substance reacts with oxygen gas, releasing energy in the form of light and heat. combustion reactions must involve o 2 as one reactant. What is a combustion reaction? a combustion reaction is an exothermic chemical reaction between substances, usually including oxygen gas and accompanied by the generation of heat, energy, and light (flame). the products of a combustion reaction depend on the combusted substance. A combustion reaction occurs when elements or compounds react with oxygen to form oxides, releasing heat in the process. it is a type of chemical reaction involving oxidation. A combustion reaction is a reaction between a fuel and oxidizer to form an oxidized product. usually, a hydrocarbon reacts with oxygen to form carbon dioxide and water. Combustion is a chemical reaction between a fuel and an oxidant that produces heat and light. the most common oxidant is oxygen, supplied through air. common fuels you'll encounter: a balanced combustion reaction has the correct stoichiometric coefficients so that atoms are conserved on both sides. for methane:. A combustion reaction is a rapid redox reaction where a substance reacts with an oxidant, usually oxygen, to produce heat and light. the reaction often produces a flame and results in the formation of oxides.
Comments are closed.