Chemical Kinetics Ncert Solved Pdf

Chemical Kinetics Ncert Solved Pdf Ans: when a temperature of rises for a chemical reaction, then the rate constant increases and becomes near to double its original value. the temperature effect on the rate constant can be represented quantitatively by the arrhenius equation. where, k = rate constant. Chemical kinetics ncert solutions provide comprehensive guidance for class 12 chemistry students preparing for cbse board examinations and competitive tests like jee main, jee advanced, and neet 2025 26.

Chemical Kinetics Ncert Solved Questions Pdf All these questions can be answered by the branch of chemistry, which deals with the study of reaction rates and their mechanisms, called chemical kinetics. the word kinetics is derived from the greek word ‘kinesis’ meaning movement. Get clear and accurate ncert solutions for class 12 chemistry chapter 3, chemical kinetics, for the academic year 2025–26, with step by step explanations and a free pdf download to help you prepare effectively for exams. Ncert exemplar for class 12 chemistry chapter 4 chemical kinetics (book solutions) free download as pdf file (.pdf), text file (.txt) or read online for free. Ncert solutions for class 12 chemistry chapter 3 chemical kinetics is study material that help students to understand the concepts related to chemical kinetics better.

Class 12th Chemistry Chemical Kinetics Ncert Solution Cbse 2023 Ncert exemplar for class 12 chemistry chapter 4 chemical kinetics (book solutions) free download as pdf file (.pdf), text file (.txt) or read online for free. Ncert solutions for class 12 chemistry chapter 3 chemical kinetics is study material that help students to understand the concepts related to chemical kinetics better. Chemical kinetics class 12 ncert solutions: aakash ncert solutions for chapter 4 of class 12 chemistry book revolves around chemical kinetics. these solutions are helpful for the students of cbse class 12th who are aspiring to make a career in the medical or engineering field. This is the 3rd chapter of chemistry for class 12th according to the revised syllabus of ncert patterns. here, all the questions of intext and exercise have been uploaded with solution in easy and simple manner. Rt solutions for chapter 4 – chemical kinetics intext questions 1. for the reaction r→pr→p, the con. entration of a reactant changes from 0.03 m to 0.02 m in 25 minutes. calculate the aver. ge rate of reaction using units of time both in minutes and seconds. ans: the average rate of the reaction can be calculated by dividing the cha. Get accurate and detailed ncert solutions for class 12 chemistry chapter 3 – chemical kinetics. understand concepts like rate of reaction, order, molecularity & more. download free pdf.
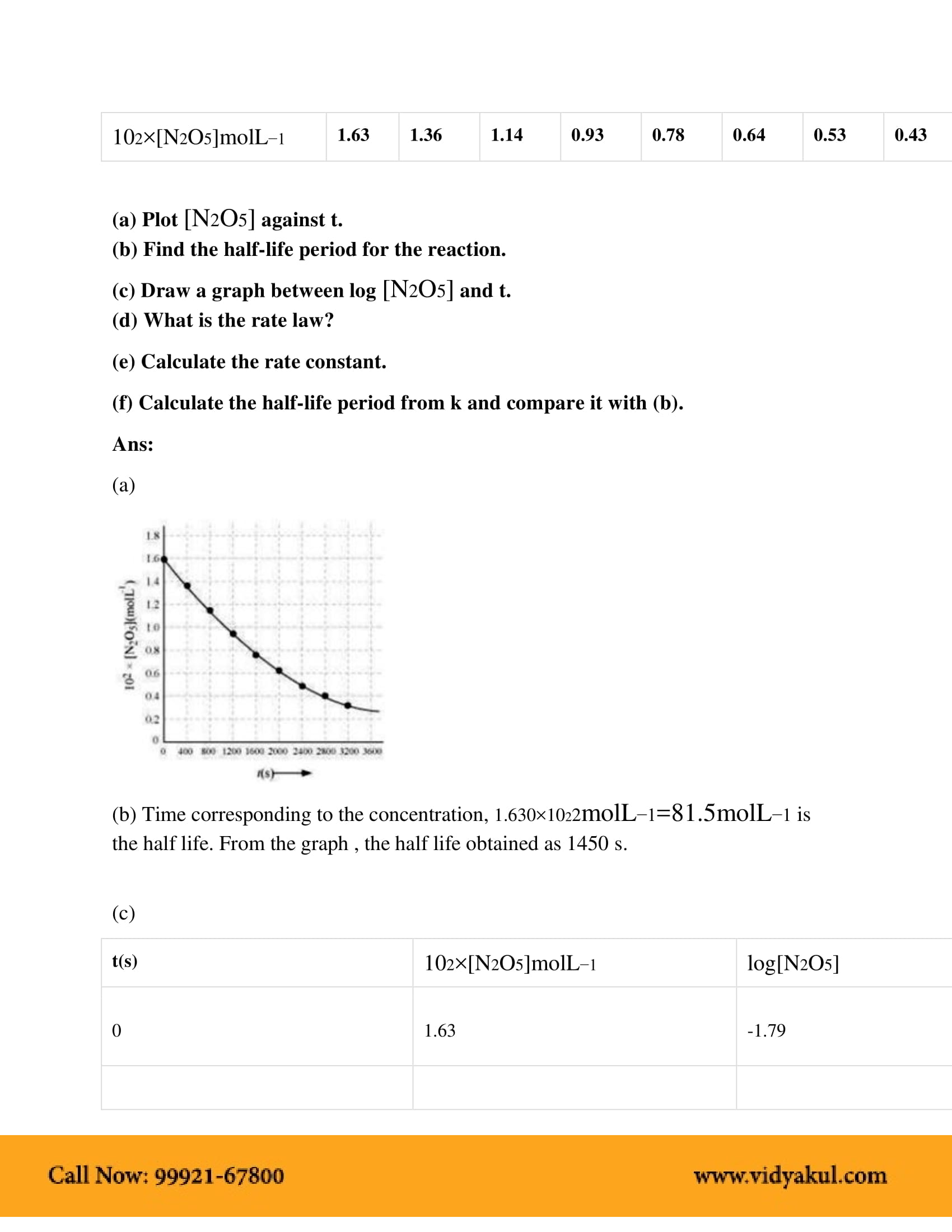
Class 12th Chemistry Chemical Kinetics Ncert Solution Cbse 2023 Chemical kinetics class 12 ncert solutions: aakash ncert solutions for chapter 4 of class 12 chemistry book revolves around chemical kinetics. these solutions are helpful for the students of cbse class 12th who are aspiring to make a career in the medical or engineering field. This is the 3rd chapter of chemistry for class 12th according to the revised syllabus of ncert patterns. here, all the questions of intext and exercise have been uploaded with solution in easy and simple manner. Rt solutions for chapter 4 – chemical kinetics intext questions 1. for the reaction r→pr→p, the con. entration of a reactant changes from 0.03 m to 0.02 m in 25 minutes. calculate the aver. ge rate of reaction using units of time both in minutes and seconds. ans: the average rate of the reaction can be calculated by dividing the cha. Get accurate and detailed ncert solutions for class 12 chemistry chapter 3 – chemical kinetics. understand concepts like rate of reaction, order, molecularity & more. download free pdf.

Class 12th Chemistry Chemical Kinetics Ncert Solution Cbse 2023 Rt solutions for chapter 4 – chemical kinetics intext questions 1. for the reaction r→pr→p, the con. entration of a reactant changes from 0.03 m to 0.02 m in 25 minutes. calculate the aver. ge rate of reaction using units of time both in minutes and seconds. ans: the average rate of the reaction can be calculated by dividing the cha. Get accurate and detailed ncert solutions for class 12 chemistry chapter 3 – chemical kinetics. understand concepts like rate of reaction, order, molecularity & more. download free pdf.
Comments are closed.