Chemical Formulas And Equations Notes

Chemical Equations Notes Pdf Chemical Reactions Hydroxide Explore writing chemical equations for igcse chemistry, using the law of conservation of mass. learn to write word and formula equations with examples. [notes: nitrogen dioxide, no2 is acidic gas and is brown in colour.].
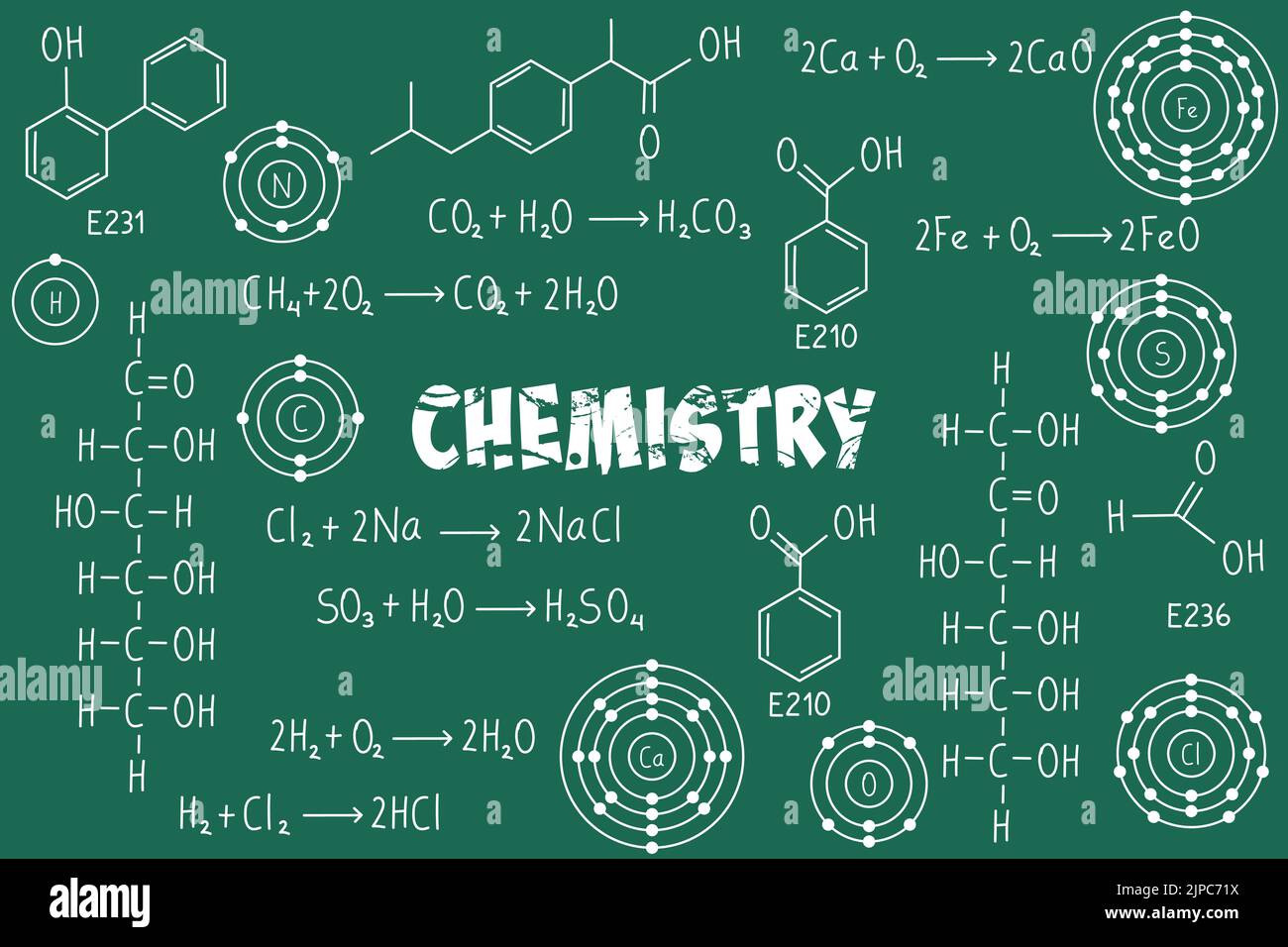
Exercise Notes Chemical Formulas And Equations Organic Chemistry Lesson notes chemical formula and chemical equations free download as pdf file (.pdf), text file (.txt) or read online for free. the document provides an overview of chemical formulas and equations, detailing types of chemical formulas such as molecular, empirical, structural, and condensed formulas, along with their significance. This activity will guide you to determine the empirical formula of a copper chloride compound using the conservation of mass, the law of definite proportions, and the law of multiple proportions. This page contains the detailed and easy notes for edexcel igcse chemistry chemical formulae,equation and calculation for revision and understanding. edexcel igcse paper 1: complete revision summary. • formation of compounds can be represented in a chemical formula. the chemical formula shows you the elements involved and the number of atoms involved. • an equation has reactants and products. reactants are substances that react together in a chemical reaction to form the products.

Exercise Notes Formulas And Equations Of Chemistry Organic Chemistry A chemical equation which is expressed by using words is called a word equation. example: carbon oxygen → carbon dioxide a chemical equation which is expressed by using symbols is called a chemical equation. example: $c (s) o 2 (g) → co 2 (g)$. To write the chemical formula for a molecule or compound, write out the chemical symbols of all of the elements in it with no spaces between them. if the molecule or compound contains more than one of a particular element, show this by putting a number to the right of that element’s symbol. All learning resources in the form of pdfs that are available on the website are 100% printable and downloadable. Chemical equation represents a chemical reaction in terms of symbols and formulae of the element and compound involved. the substance or ions taking part in the reaction, i.e. reactants are written on the left hand side while the substance formed in the reaction product are written on the right hand side.
Comments are closed.