Chem110 Practice Exam 2 Pdf

Chem110 Practice Exam 2 Pdf Chem110 practice exam 2 free download as pdf file (.pdf), text file (.txt) or read online for free. 30) what are the coefficients of iron (fe), phosphoric acid (h3po4), iron(ii) phosphate (fe3(po4)2) and hydrogen (h2) when the following equation is balanced?.

Chem110 Practice Exam 1 Pdf Periodic Table Atoms Once the exam period is over, your work will be uploaded to the “exam 2” assignment under the modules tab on canvas. this exam is worth 100 points (max possible 102). the exam period is 60 minutes. at the end of 60 minutes, you will stop working and submit your written work to the assignment in canvas. any explanations should be succinct. When 16 g of methane is burned according to the equation ch 4 2o 2 co 2 2h 2 o , you experimentally measure you have produced 32 g of water. what is the percent yield for this reaction?. Part 1 exam 2 (last) (first) 1. draw the lewis dot structure for each of the following (12 points). Level up your studying with ai generated flashcards, summaries, essay prompts, and practice tests from your own notes. sign up now to access chem110 exam 2 practice materials and ai powered study resources.

Chem110 Practice Exam 1 Pdf Periodic Table Atoms Part 1 exam 2 (last) (first) 1. draw the lewis dot structure for each of the following (12 points). Level up your studying with ai generated flashcards, summaries, essay prompts, and practice tests from your own notes. sign up now to access chem110 exam 2 practice materials and ai powered study resources. Once you begin the quiz, you have 50 minutes to complete it. a maximum of three attempts are allowed. you are welcome to print the quiz the first time you take it, prior to submitting it (so you have a record of the questions.) during the actual exam, only one question will be visible at a time. Chem110 answer to practice exam 2 1) b 2) d 3) d 4) e 5) a 6) a 7) b 8) b 9) a 10) e 11) e 12) b 13) c 14) a 15) d 16) b 17) e. Avogadro's number na = 6.022 x 1023 = 1 mole soluble compounds compounds containing alkali metals ions and the ammonium ion nitrates, bicarbonates, and chlorates halides sulfates insoluble compounds carbonates, phosphates, chromates, and sulfides hydroxides exceptions halides of ag , hg22 , and pb2 sulfates of ag , ca2 , sr2 , ba2 , hg22 , and pb2 exceptions compounds containing alkali metals ions and the ammonium ion compounds containing alkali metals ions and the ammonium ion 2 choose the most appropriate answer. Stanich chem110 practiceexam2 free download as pdf file (.pdf), text file (.txt) or view presentation slides online. the document is a practice exam for chem 110 a, covering chapters 12, 5, and 8, with a total of 12 questions to be completed in 45 minutes.

Chem110 Practice Exam 1 Pdf Periodic Table Atoms Once you begin the quiz, you have 50 minutes to complete it. a maximum of three attempts are allowed. you are welcome to print the quiz the first time you take it, prior to submitting it (so you have a record of the questions.) during the actual exam, only one question will be visible at a time. Chem110 answer to practice exam 2 1) b 2) d 3) d 4) e 5) a 6) a 7) b 8) b 9) a 10) e 11) e 12) b 13) c 14) a 15) d 16) b 17) e. Avogadro's number na = 6.022 x 1023 = 1 mole soluble compounds compounds containing alkali metals ions and the ammonium ion nitrates, bicarbonates, and chlorates halides sulfates insoluble compounds carbonates, phosphates, chromates, and sulfides hydroxides exceptions halides of ag , hg22 , and pb2 sulfates of ag , ca2 , sr2 , ba2 , hg22 , and pb2 exceptions compounds containing alkali metals ions and the ammonium ion compounds containing alkali metals ions and the ammonium ion 2 choose the most appropriate answer. Stanich chem110 practiceexam2 free download as pdf file (.pdf), text file (.txt) or view presentation slides online. the document is a practice exam for chem 110 a, covering chapters 12, 5, and 8, with a total of 12 questions to be completed in 45 minutes.
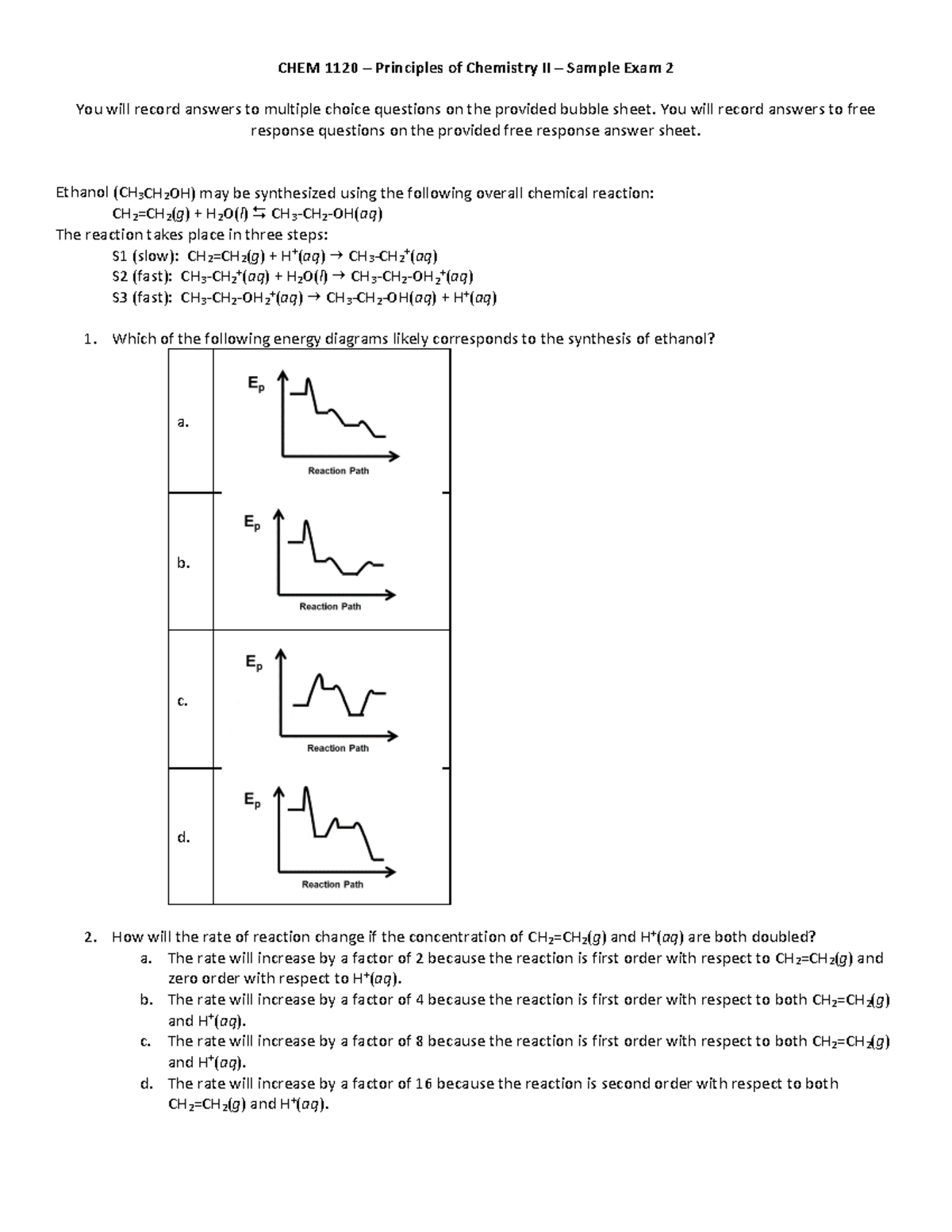
Chem 1120 Sample Exam 2 Practice Questions And Solutions Studocu Avogadro's number na = 6.022 x 1023 = 1 mole soluble compounds compounds containing alkali metals ions and the ammonium ion nitrates, bicarbonates, and chlorates halides sulfates insoluble compounds carbonates, phosphates, chromates, and sulfides hydroxides exceptions halides of ag , hg22 , and pb2 sulfates of ag , ca2 , sr2 , ba2 , hg22 , and pb2 exceptions compounds containing alkali metals ions and the ammonium ion compounds containing alkali metals ions and the ammonium ion 2 choose the most appropriate answer. Stanich chem110 practiceexam2 free download as pdf file (.pdf), text file (.txt) or view presentation slides online. the document is a practice exam for chem 110 a, covering chapters 12, 5, and 8, with a total of 12 questions to be completed in 45 minutes.
Comments are closed.