Chem Lab 4 Solid Liquid Gas
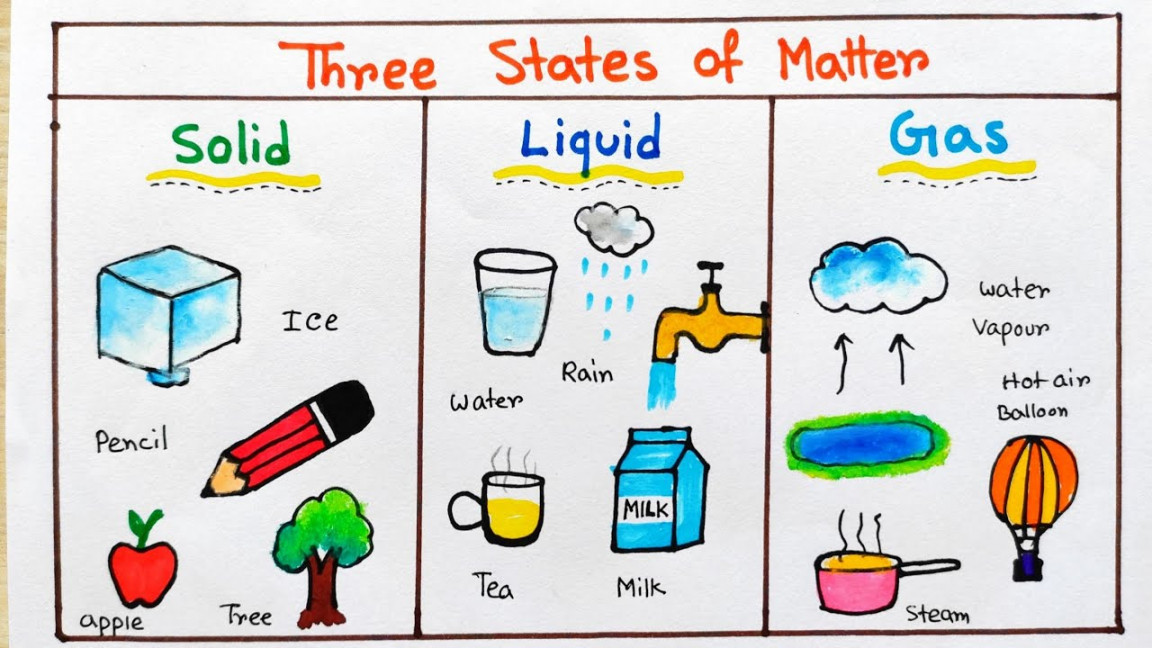
Pictures Of Solid Liquid And Gas Objects Printable Pdf Template This page explores the phases of matter—solid, liquid, and gas—highlighting the influence of intermolecular interactions on their characteristics. strong interactions define solids, while weaker ones in liquids and gases enable flow and expansion. Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on .

Solid Liquid And Gas Posters Made By Teachers Heat, cool and compress atoms and molecules and watch as they change between solid, liquid and gas phases. Discover how solids, liquids and gases differ in chemistry. learn how particle behavior defines each state and see real‑world examples. Students explore the attractions and motion of atoms and molecules as they experiment with and observe the heating and cooling of a solid, liquid, and gas. a summary of the investigations and science content from all lessons in the chapter. evaluate students’ progress. When the disruptive forces are greater than the cohesive forces, we can have a change of state (e. liquid → gas). it takes energy to make this transition, and since the energy is used to evaporate the molecules, the remaining liquid gets cooler.
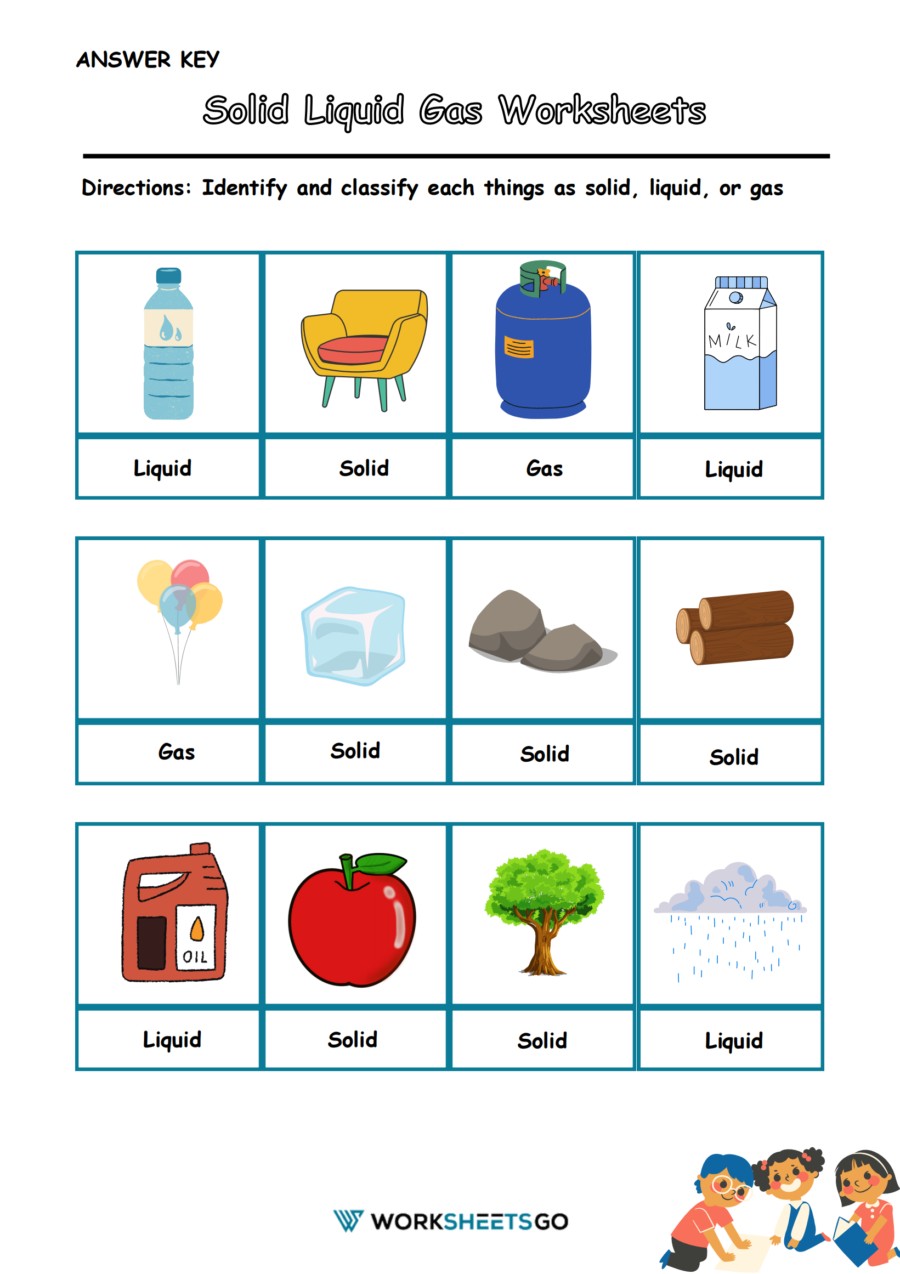
Solid Liquid Gas Worksheets Worksheetsgo Students explore the attractions and motion of atoms and molecules as they experiment with and observe the heating and cooling of a solid, liquid, and gas. a summary of the investigations and science content from all lessons in the chapter. evaluate students’ progress. When the disruptive forces are greater than the cohesive forces, we can have a change of state (e. liquid → gas). it takes energy to make this transition, and since the energy is used to evaporate the molecules, the remaining liquid gets cooler. This video takes a look at the molecular structure of solids, liquids, and gases and examines how the kinetic energy of the particles changes. the video also discusses melting, vaporization, condensation, and freezing. Let’s put together and discuss some of the general properties characteristic of solids, liquids, and gases. we have talked about the shape of solids, liquids, and gases. * * * some writing from kotz (powerpoint online) solid, liquid, gas are the three states of matter we will deal with. plasma and neutron star are also states of matter. Watch different types of molecules form a solid, liquid, or gas. add or remove heat and watch the phase change. change the temperature or volume of a container and see a pressure temperature diagram respond in real time. relate the interaction potential to the forces between molecules.
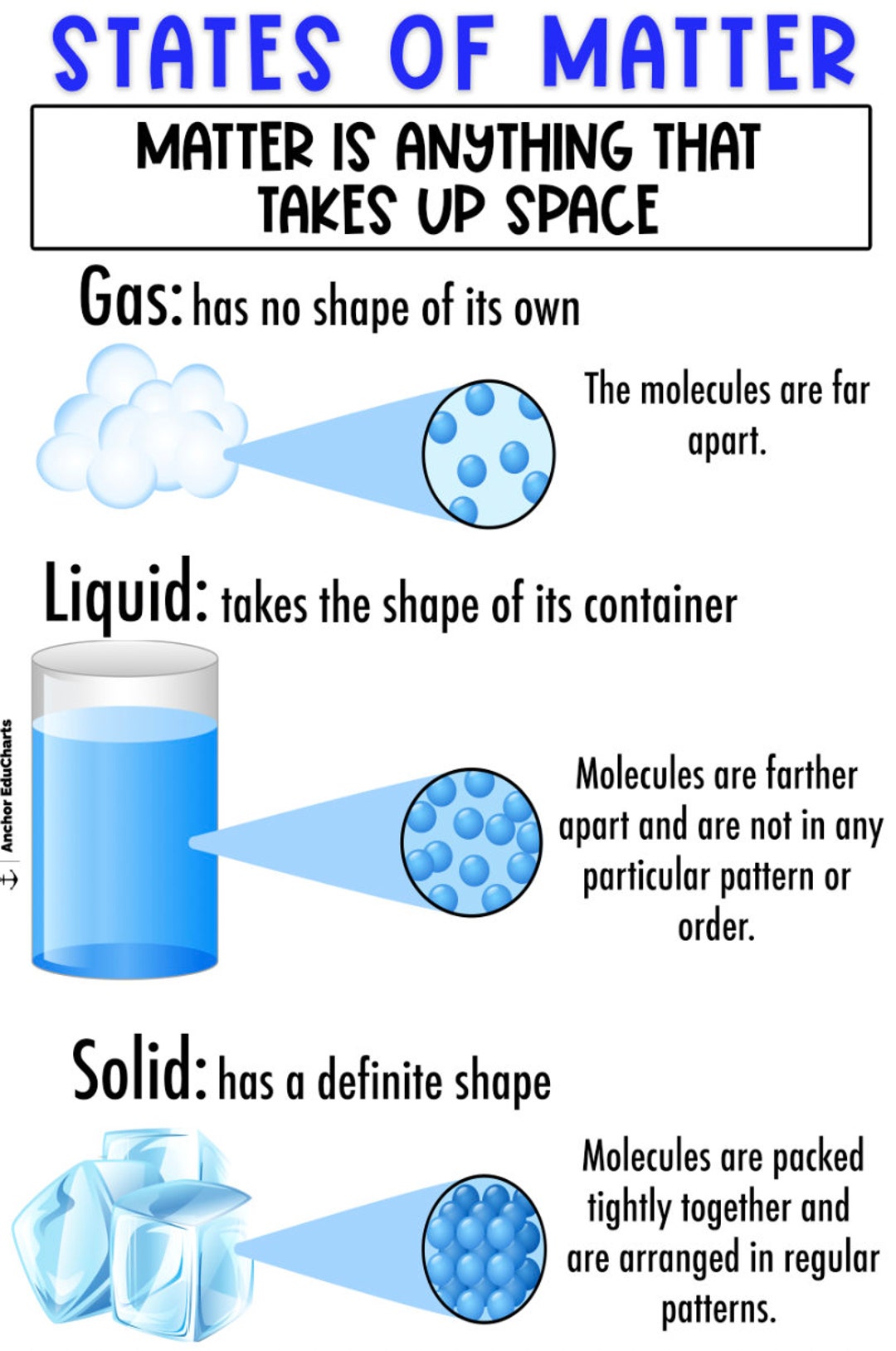
Solid Liquid Gas Chart Investigate The Movement Of Particles In This video takes a look at the molecular structure of solids, liquids, and gases and examines how the kinetic energy of the particles changes. the video also discusses melting, vaporization, condensation, and freezing. Let’s put together and discuss some of the general properties characteristic of solids, liquids, and gases. we have talked about the shape of solids, liquids, and gases. * * * some writing from kotz (powerpoint online) solid, liquid, gas are the three states of matter we will deal with. plasma and neutron star are also states of matter. Watch different types of molecules form a solid, liquid, or gas. add or remove heat and watch the phase change. change the temperature or volume of a container and see a pressure temperature diagram respond in real time. relate the interaction potential to the forces between molecules.

Solid Liquid Gas Worksheet Worksheets Library * * * some writing from kotz (powerpoint online) solid, liquid, gas are the three states of matter we will deal with. plasma and neutron star are also states of matter. Watch different types of molecules form a solid, liquid, or gas. add or remove heat and watch the phase change. change the temperature or volume of a container and see a pressure temperature diagram respond in real time. relate the interaction potential to the forces between molecules.
Comments are closed.