Charles Law Demonstration

3 1 Charles Law Demonstration 1 Demonstration 2 Method Pdf Bottle A demonstration of charles' law done by heating up a balloon while under constant pressure. Charles's law states that the volume of an ideal gas changes proportionally to the temperature of that gas, given that pressure and amount of gas present are held constant.
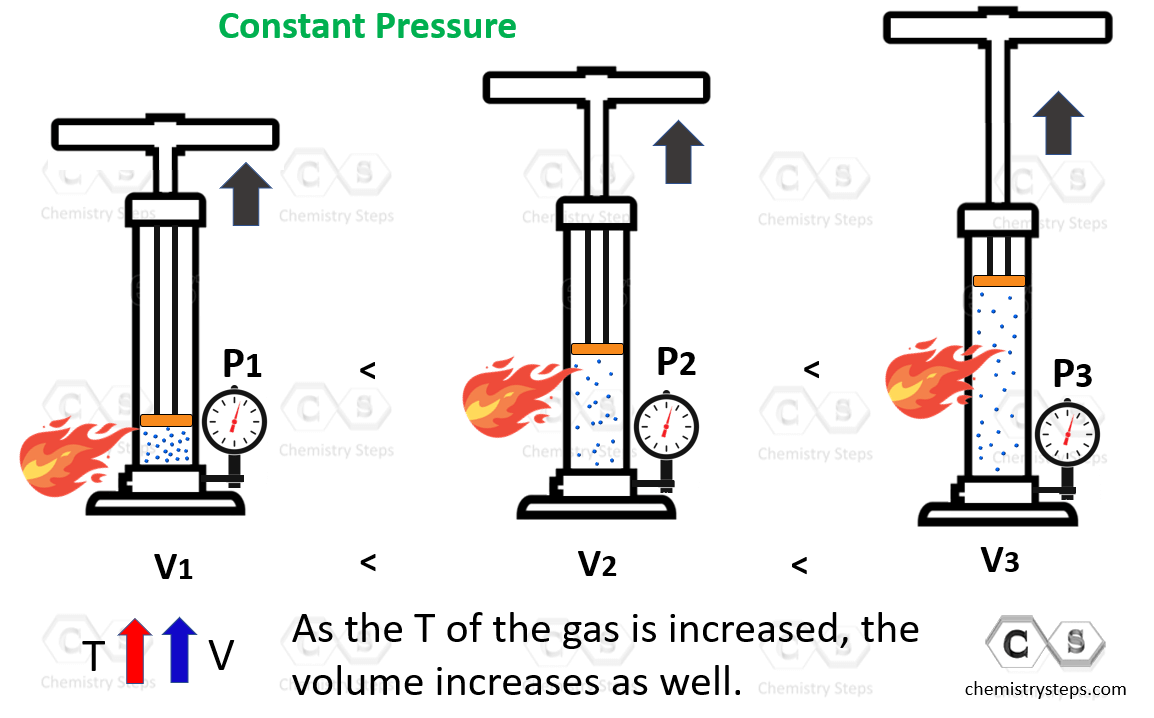
Charle S Law Chemistry Steps Explore charles's law with a visual experiment using a balloon and temperature changes. ideal for students and science enthusiasts alike. Charles' law predicts a fourfold decrease in the volume of a gas as it is chilled from room temperature, 20 degrees celsius, to liquid nitrogen temperature, 196 degrees celsius. if the gas in the balloon condenses, as oxygen gas does at 183 c, the volume change is much greater. Demonstrate charles’s law can crushing experiment. observe how temperature changes the volume of gas for a fun, hands on science lesson. Learn about jacques charles's experiments with gas and temperature, and discover how heating a gas in a closed container under constant pressure increases its volume.

Charles S Law Demonstration Easy Visual Educational Experiment Demonstrate charles’s law can crushing experiment. observe how temperature changes the volume of gas for a fun, hands on science lesson. Learn about jacques charles's experiments with gas and temperature, and discover how heating a gas in a closed container under constant pressure increases its volume. Several experiments can elegantly demonstrate the behavior described by charles’s law. in this article, we will explore three ways to demonstrate charles’s law in a simple and accessible manner. As the flask is warmed from 0ºc to room temperature (25°c), the gas expands inside the flask and slowly moves the syringe plunger. this demonstration demonstrates the relationship between temperature and volume (charles' law), and can be used to extrapolate absolute zero. contributors dr. george bodner (perdue university). This demonstration demonstrates the relationship between temperature and volume (charles' law), and can be used to extrapolate absolute zero. Educators: to receive phet's monthly newsletter, register for a free educator account. ©2026 university of colorado. some rights reserved.

Charles Law Balloon Over 11 Royalty Free Licensable Stock Vectors Several experiments can elegantly demonstrate the behavior described by charles’s law. in this article, we will explore three ways to demonstrate charles’s law in a simple and accessible manner. As the flask is warmed from 0ºc to room temperature (25°c), the gas expands inside the flask and slowly moves the syringe plunger. this demonstration demonstrates the relationship between temperature and volume (charles' law), and can be used to extrapolate absolute zero. contributors dr. george bodner (perdue university). This demonstration demonstrates the relationship between temperature and volume (charles' law), and can be used to extrapolate absolute zero. Educators: to receive phet's monthly newsletter, register for a free educator account. ©2026 university of colorado. some rights reserved.
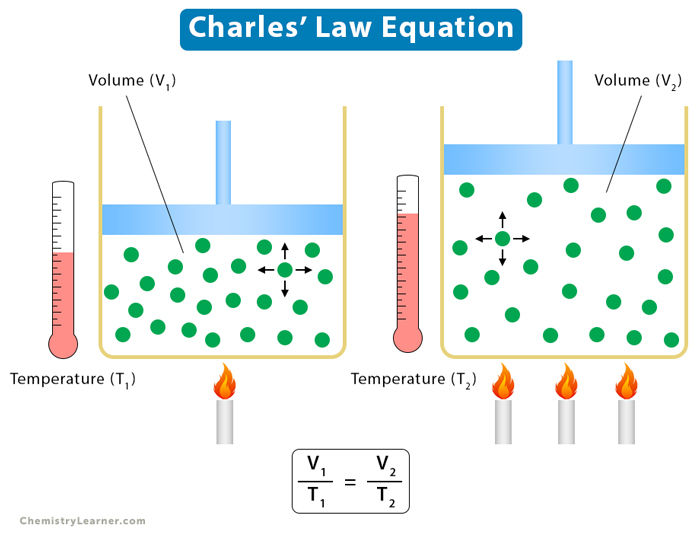
Charles Law Statement Formula Examples And Graph This demonstration demonstrates the relationship between temperature and volume (charles' law), and can be used to extrapolate absolute zero. Educators: to receive phet's monthly newsletter, register for a free educator account. ©2026 university of colorado. some rights reserved.
Comments are closed.