Charge Balance Equations Chemistry Mono Mole

Lecture 2 Mole Balance Equations Pdf Chemical Reactor Reaction Rate Charge balance equations are derived using the concept of electroneutrality, where the sum of positive charges equals to the sum of negative charges in a solution. such equations are useful for analysing acid base equilibria and formulating complex acid base titration equations. This equation is known as a charge balance. it is important to realize that all solutions must be electrically neutral; that is, for every substance of positive charge there must be an equivalent amount of negative charge to balance it out.

Balancing Chemical Equations Pdf Mole Unit Chemistry Lecture notes covering complex equilibria, edta titrations, and electroanalytical chemistry principles. includes charge & mass balance equations. The charge balance equation equates the total positive charge to the total negative charge, taking into account all dissociating species including water. several examples are provided of writing the mass balance and charge balance equations for solutions containing sodium salts. Let’s go back and write a charge balance for the nahpo4. in many instances when we are dealing with very complex equilibria, it is useful to use mass balance and charge balance in efforts to reduce an expression to one variable. we will use these in chapter 11 and 12, and then again when we come back to chapter 9. chapter 10 diprotic acids. Understand the core concept of electrical neutrality and learn practical methods for balancing charges in compounds and chemical equations.
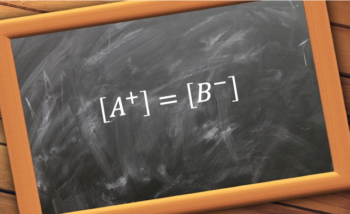
Charge Balance Equations Chemistry Mono Mole Let’s go back and write a charge balance for the nahpo4. in many instances when we are dealing with very complex equilibria, it is useful to use mass balance and charge balance in efforts to reduce an expression to one variable. we will use these in chapter 11 and 12, and then again when we come back to chapter 9. chapter 10 diprotic acids. Understand the core concept of electrical neutrality and learn practical methods for balancing charges in compounds and chemical equations. Check ionic electroneutrality with clear charge totals. convert common units safely. spot sampling errors fast with a balance percentage. 11a 2 charge balance equation for any solution containing electrolytes, no. moles l positive charge = no. moles l negative charge this equation represents the charge balance condition and is called the charge balance equation. First step: write total ionic equations for everything involved. note that "1%" means "a very small amount" and that "99%" means most of the agcl stays undissociated. Charge is balanced in oxidation half reactions by adding electrons as products; in reduction half reactions, charge is balanced by adding electrons as reactants.

Charge Balance Equations Chemistry Mono Mole Check ionic electroneutrality with clear charge totals. convert common units safely. spot sampling errors fast with a balance percentage. 11a 2 charge balance equation for any solution containing electrolytes, no. moles l positive charge = no. moles l negative charge this equation represents the charge balance condition and is called the charge balance equation. First step: write total ionic equations for everything involved. note that "1%" means "a very small amount" and that "99%" means most of the agcl stays undissociated. Charge is balanced in oxidation half reactions by adding electrons as products; in reduction half reactions, charge is balanced by adding electrons as reactants.
Charge Balance Equations Chemistry Mono Mole First step: write total ionic equations for everything involved. note that "1%" means "a very small amount" and that "99%" means most of the agcl stays undissociated. Charge is balanced in oxidation half reactions by adding electrons as products; in reduction half reactions, charge is balanced by adding electrons as reactants.
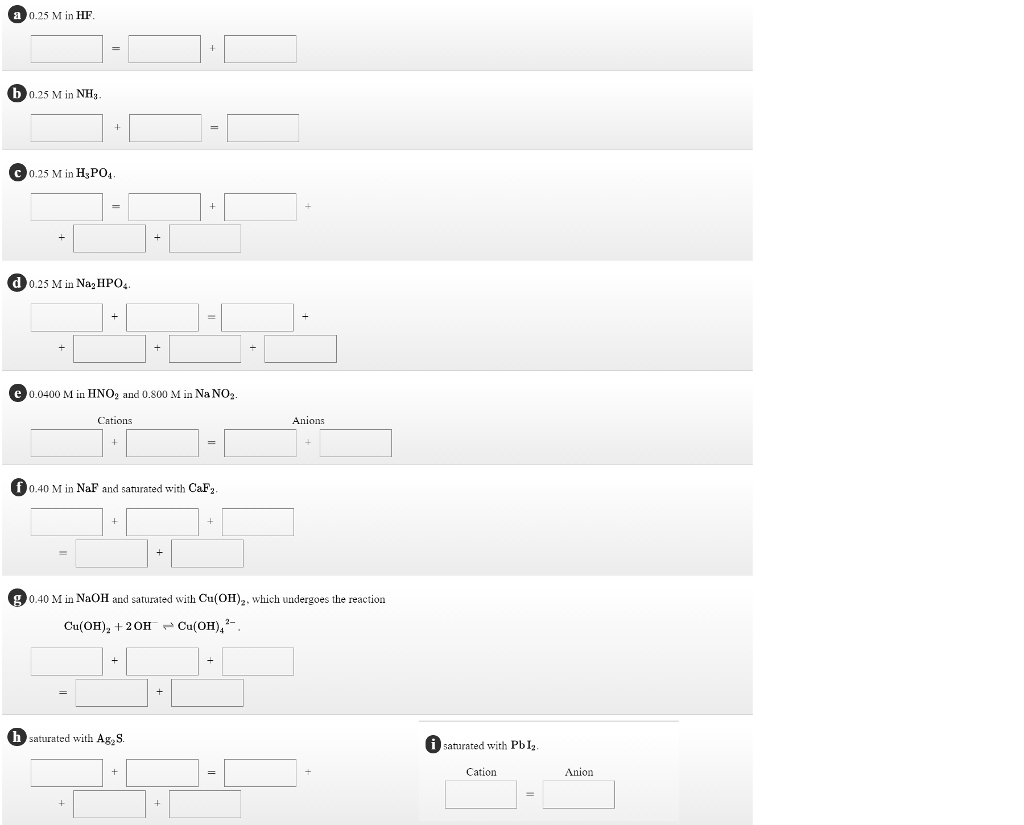
Solved Write The Charge Balance Equations For A Solution Chegg
Comments are closed.