Chapter 7 Quantum Two Body Problem Hydrogen Atom

Chapter 7 Quantum Two Body Problem Hydrogen Atom Explore the interaction between spinless point particles in 3d, their operators, and commutation relations. study hamiltonians, transformations, and central potential behavior. Mit opencourseware is a web based publication of virtually all mit course content. ocw is open and available to the world and is a permanent mit activity.

Chapter 7 Quantum Two Body Problem Hydrogen Atom In quantum mechanics, this 2 body system is described by a hamiltonian of the form. for simplicity, we assume both particles are spinless. x1 ! x1 a; x2 ! x2 a; and that's why the 2 particles interact with each other but are not subject to any external forces. Chapter 7 the hydrogen atom before discussing the h atom solutions to the schr ̈odinger equation, however, we start with a general discussion of the dynamic of 2 body systems in both classical and quantum me chanics. Since the two interactions are attractive, bound states are expected both for the hydrogen atom and for the deuteron nucleus. in fact, both of them exist in nature. The dutch physicist pieter zeeman showed the spectral lines emitted by atoms in a magnetic field split into multiple energy levels. it is called the zeeman effect.

Chapter 7 Quantum Two Body Problem Hydrogen Atom Since the two interactions are attractive, bound states are expected both for the hydrogen atom and for the deuteron nucleus. in fact, both of them exist in nature. The dutch physicist pieter zeeman showed the spectral lines emitted by atoms in a magnetic field split into multiple energy levels. it is called the zeeman effect. In 1927, heisenberg described the problem by stating it is inherently impossible to know simultaneously both the exact momentum (mv) of the electron and its exact location (x) in space. In this chapter we will be studying as illustrative examples of the application of the schr ̈o dinger equation, the hydrogen atom problem and the one dimensional linear harmonic oscillator. From chapter 4, we obtained a general result for the wave function of an electron bound to the nucleus of a hydrogen atom, which allowed us to write the energy of such states as follows;\\. Consider a system of two particles with no external fields by symmetry, the interaction energy can only depend on the separation distance:.
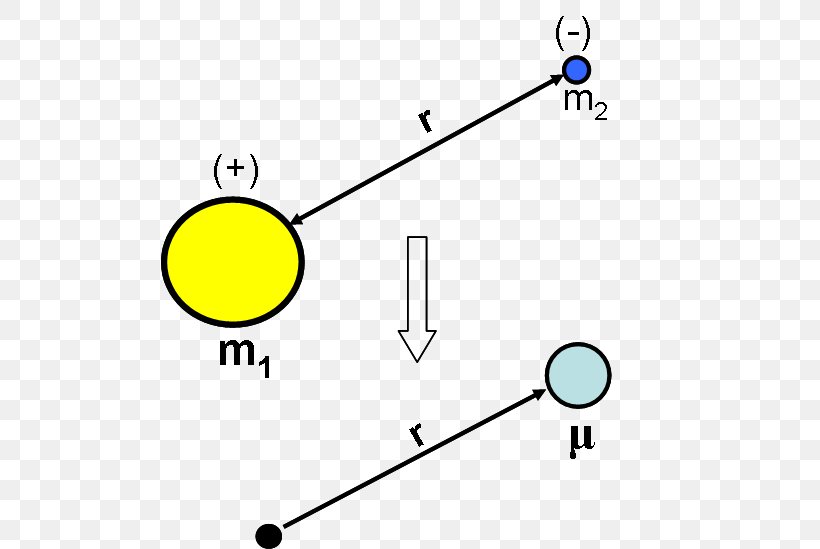
Reduced Mass Two Body Problem Hydrogen Atom Schrödinger Equation In 1927, heisenberg described the problem by stating it is inherently impossible to know simultaneously both the exact momentum (mv) of the electron and its exact location (x) in space. In this chapter we will be studying as illustrative examples of the application of the schr ̈o dinger equation, the hydrogen atom problem and the one dimensional linear harmonic oscillator. From chapter 4, we obtained a general result for the wave function of an electron bound to the nucleus of a hydrogen atom, which allowed us to write the energy of such states as follows;\\. Consider a system of two particles with no external fields by symmetry, the interaction energy can only depend on the separation distance:.

Reduced Mass Two Body Problem Hydrogen Atom Schrödinger Equation From chapter 4, we obtained a general result for the wave function of an electron bound to the nucleus of a hydrogen atom, which allowed us to write the energy of such states as follows;\\. Consider a system of two particles with no external fields by symmetry, the interaction energy can only depend on the separation distance:.
Comments are closed.