Chapter 5 Part 1 The Diatomic Elements

Chapter 5 Part 1 Pdf Chapter 5 – part 1: the diatomic elements chemistry unleashed 38.2k subscribers subscribed. There are seven diatomic elements, aka molecular elements, all listed here. learn about what a diatomic element is and how it's different from a diatomic molecule.

Diatomic Elements Diagram Quizlet Diatomic elements are pure elements that form molecules consisting of two atoms bonded together. there are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, and bromine. While some elements exist as molecules, most elements exist with individual atoms as their basic unit. if an element is not one of those listed above as a molecular element, assume it is an atomic element. Lecture notes for chapter 5 of intro chem. chapter gases elements and molecules elements noble gases and diatomic all never ionic compounds molecular compounds. A diatomic molecule is a molecule containing only two atoms. there are seven diatomic elements that you have to remember and a simple mnemonic to help with this.
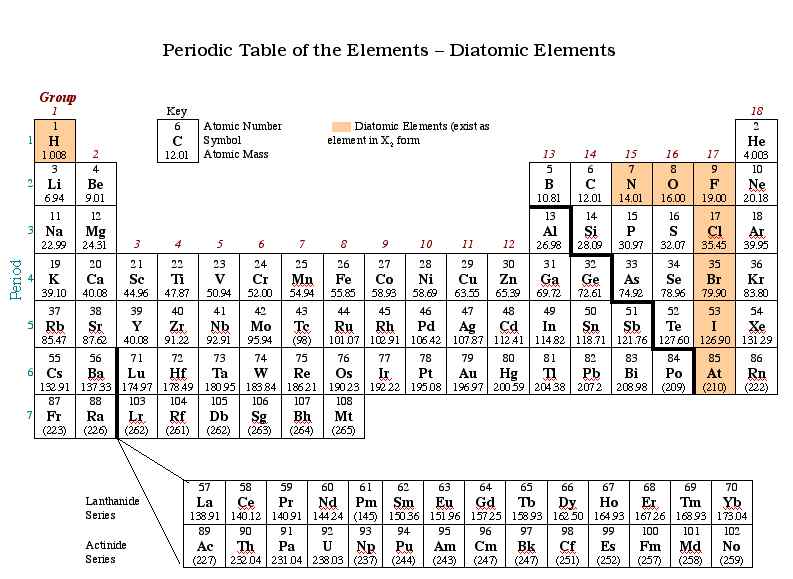
1b6 Diatomic Elements Lecture notes for chapter 5 of intro chem. chapter gases elements and molecules elements noble gases and diatomic all never ionic compounds molecular compounds. A diatomic molecule is a molecule containing only two atoms. there are seven diatomic elements that you have to remember and a simple mnemonic to help with this. Learn test blocks match hydrogen h2 1 7 1 7 created by sageamelia93 created 2 years ago. The total energy is the sum of (5.30) and (5.31). this can be written out explicitly writing s from (5.24) in the following form: total energy = ke pe = α2f1(αr) αf2(αr). Video answers for all textbook questions of chapter 5, heteronuclear diatomic molecules, chemistry by numerade. This is a list of all of the diatomic elements and their common properties. simple mnemonics for remembering them are included.
Comments are closed.