Changes To Orange Book Patent Information

1987 Orange Book Pdf Transparency act (obta) of 2020 (pub. l. 116 290). section 2(e) of the obta requires the agency (1) to solicit public comments regarding the types of patent information that should be included. Although the first draft edition (1979) and the current 45 th annual edition (2025) are fundamentally similar in their goals, the orange book is constantly in flux, with new product information (e.g., approvals, patents) posted daily and many other substantive updates made over its 45‑year history.

Orange Book 2nd Edition 2011 Addendum Pdf Interest Employment Congress updated the statute on the types of patents that should be listed in 2021 (p.l. 116 290), and fda has also issued and updated its regulations on these issues. even so, disputes continue over whether certain patent types (e.g., rems or drug delivery devices) should be listed. Orange book users should not rely on an orange book patent listing to identify the complete range of patent claims that may be asserted by an nda holder or patent owner. The orange book transparency act (obta), enacted as part of the consolidated appropriations act, 2021, significantly updated the drug approval and patent listing process. Until fda takes steps to clarify listing requirements for certain rems and medical device patents, heightened scrutiny is required when evaluating the eligibility of such patents for listing in the orange book, including the validity of existing listings.

Download The Complete Fda Orange Book Foia Archives From 1980 Present The orange book transparency act (obta), enacted as part of the consolidated appropriations act, 2021, significantly updated the drug approval and patent listing process. Until fda takes steps to clarify listing requirements for certain rems and medical device patents, heightened scrutiny is required when evaluating the eligibility of such patents for listing in the orange book, including the validity of existing listings. Delistings, patent corrections, and supplements: monitor changes and predict the patent cliff in the fda orange book by drugpatentwatch february 28, 2026. We are responding to a request for comments on the types of patents currently listed in the orange book and the impact that any change to current patent listing practices may have on drug product development. Fda has prepared this report, entitled report to congress: the listing of patent information in the orange book, 5 which summarizes the public comments received regarding the types of patent information that should be included in, or removed from, the orange book. Recent court rulings have changed the way branded and generic drug manufacturers should think about patent listings in the fda orange book.

Ftc Orange Book Patent Challenges Rxdatalab Delistings, patent corrections, and supplements: monitor changes and predict the patent cliff in the fda orange book by drugpatentwatch february 28, 2026. We are responding to a request for comments on the types of patents currently listed in the orange book and the impact that any change to current patent listing practices may have on drug product development. Fda has prepared this report, entitled report to congress: the listing of patent information in the orange book, 5 which summarizes the public comments received regarding the types of patent information that should be included in, or removed from, the orange book. Recent court rulings have changed the way branded and generic drug manufacturers should think about patent listings in the fda orange book.

Fda Orange Book Pdf Library With Historical Archives From 1980 2025 Fda has prepared this report, entitled report to congress: the listing of patent information in the orange book, 5 which summarizes the public comments received regarding the types of patent information that should be included in, or removed from, the orange book. Recent court rulings have changed the way branded and generic drug manufacturers should think about patent listings in the fda orange book.
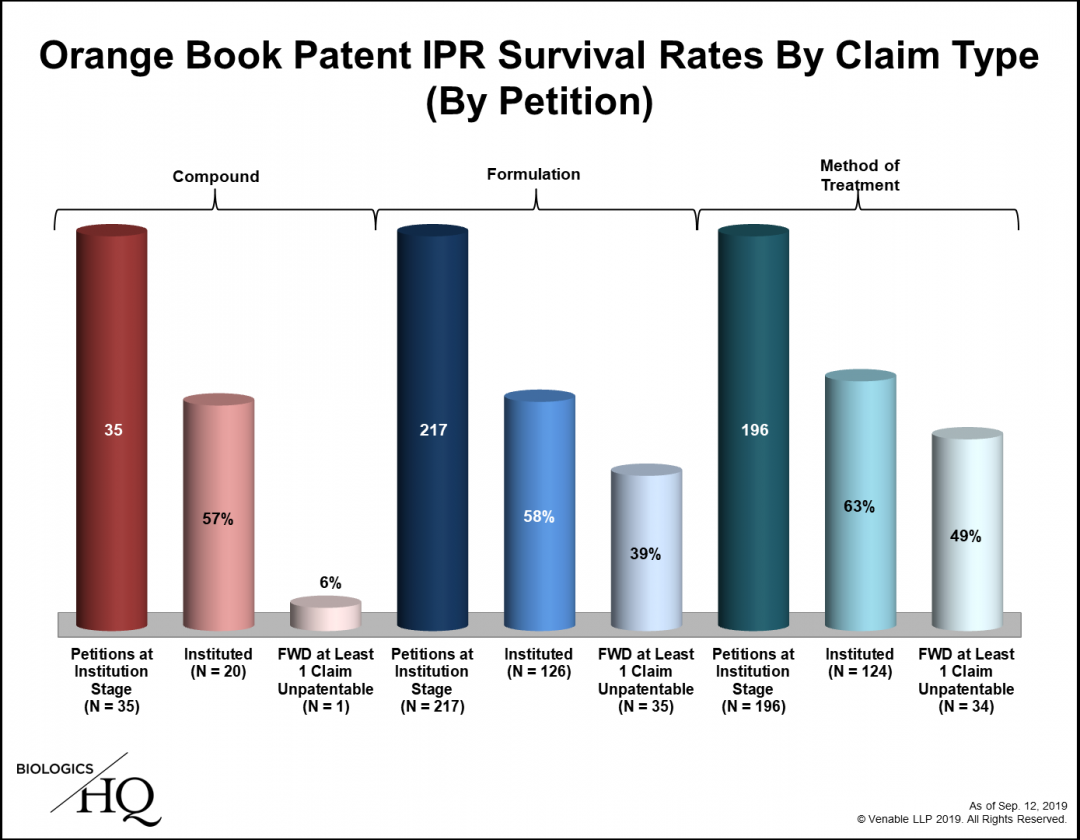
Seven Years Of Orange Book Patent Iprs Where Are We Now Venable S
Comments are closed.