Catalyst

Catalyst Catalyst, in chemistry, any substance that increases the rate of a reaction without itself being consumed. enzymes are naturally occurring catalysts responsible for many essential biochemical reactions. In general, chemical reactions occur faster in the presence of a catalyst because the catalyst provides an alternative reaction mechanism (reaction pathway) having a lower activation energy than the noncatalyzed mechanism.

Labflow By donating to catalyst or enabling corporate matching, you invest in research that drives cultural transformation, convenings that accelerate leadership, and proven solutions that embed inclusion into business practice at scale. 972.999.0081 email info@catalystcommercial at catalyst we build high impact solutions that can create “complete communities.”. Learn about the types, mechanisms and applications of catalysts in chemistry. find out how catalysts can change the activation energy, rate and selectivity of chemical reactions, and how they can be poisoned or promoted by other substances. Find out about catalytic reaction & catalyst. check out its characteristics, types, examples, kinetics, & mechanism. learn catalyzed vs. uncatalyzed reactions.
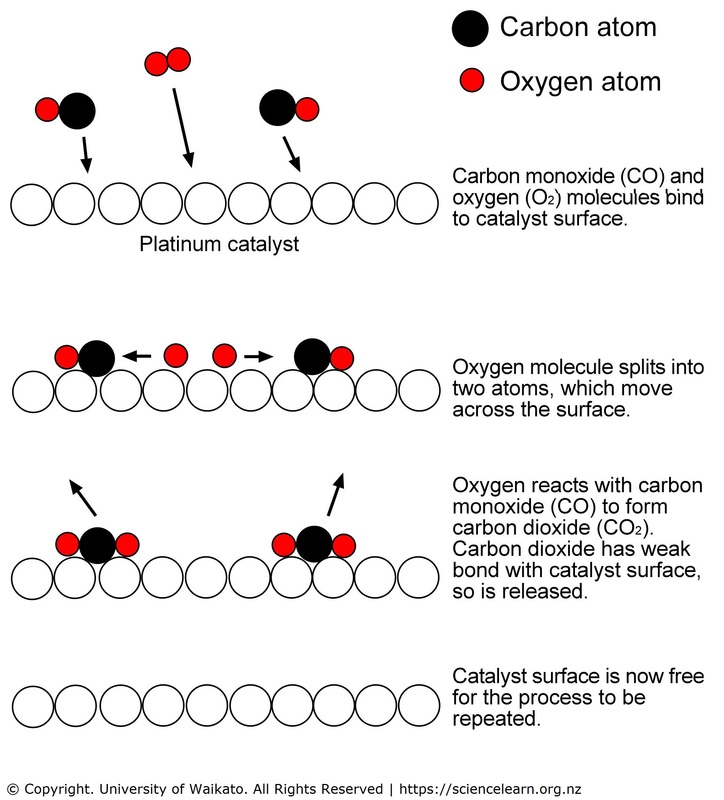
Catalytic Converter Catalyst Science Learning Hub Learn about the types, mechanisms and applications of catalysts in chemistry. find out how catalysts can change the activation energy, rate and selectivity of chemical reactions, and how they can be poisoned or promoted by other substances. Find out about catalytic reaction & catalyst. check out its characteristics, types, examples, kinetics, & mechanism. learn catalyzed vs. uncatalyzed reactions. A catalyst in chemistry is a substance that accelerates a chemical reaction by offering an alternative pathway with lower activation energy. this action enhances the reaction rate without being consumed or undergoing permanent alteration. A catalyst enhances the rate of a reaction. in other words, they allow a higher fraction of molecules to reach the minimum energy required for the reaction; hence, leading to the formation of more products. In principal, a catalyst aids transformation of its products while remaining unchanged itself, supporting indefinite reuse. in practice, secondary reactions gradually consume most industrial catalysts, making it important to avoid expensive agents. Each reaction occurs at a certain rate, which can be altered by adding a material called a catalyst. the catalyst is neither a reactant nor a product; it does not get consumed during the reaction.

Unveiling The Inner Workings A Visual Guide To A Catalyst A catalyst in chemistry is a substance that accelerates a chemical reaction by offering an alternative pathway with lower activation energy. this action enhances the reaction rate without being consumed or undergoing permanent alteration. A catalyst enhances the rate of a reaction. in other words, they allow a higher fraction of molecules to reach the minimum energy required for the reaction; hence, leading to the formation of more products. In principal, a catalyst aids transformation of its products while remaining unchanged itself, supporting indefinite reuse. in practice, secondary reactions gradually consume most industrial catalysts, making it important to avoid expensive agents. Each reaction occurs at a certain rate, which can be altered by adding a material called a catalyst. the catalyst is neither a reactant nor a product; it does not get consumed during the reaction.
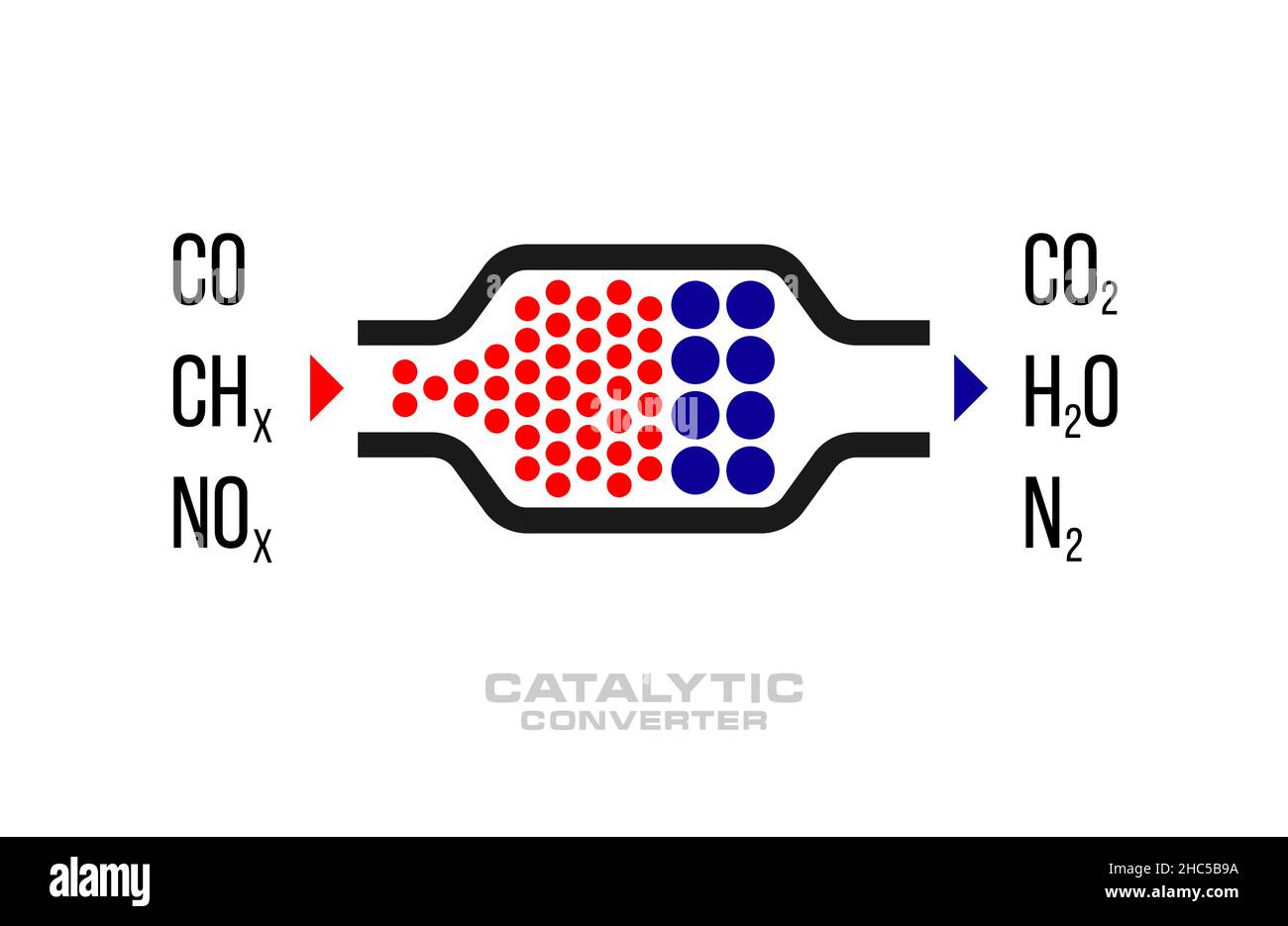
Catalyst Diagram Chemistry At Mary Galvin Blog In principal, a catalyst aids transformation of its products while remaining unchanged itself, supporting indefinite reuse. in practice, secondary reactions gradually consume most industrial catalysts, making it important to avoid expensive agents. Each reaction occurs at a certain rate, which can be altered by adding a material called a catalyst. the catalyst is neither a reactant nor a product; it does not get consumed during the reaction.

Contact Catalyst
Comments are closed.