Calcium Chloride Water

Calcium Chloride 90 Syntech Calcium chloride is an inorganic compound, a salt with the chemical formula cacl2. it is a white crystalline solid at room temperature, and it is highly soluble in water. it can be created by neutralising hydrochloric acid with calcium hydroxide. Learn why calcium chloride is in your water, distinguishing between natural sources and intentional use for taste, safety, and pipe health.
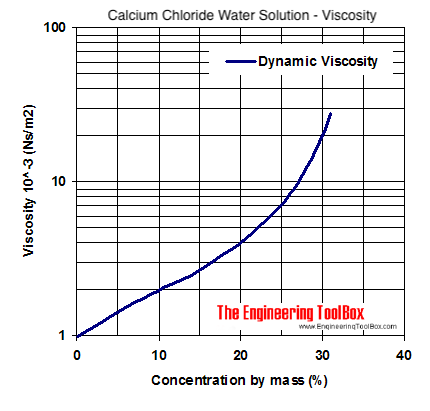
Calcium Chloride In Water This article explains the mechanism of calcium chloride and water reactions and also lists their applications, linking their molecular structure with the topic. Calcium chloride is an ionic compound composed of calcium ions (ca²⁺) and chloride ions (cl⁻). its high solubility in water is a key factor in its widespread use. ionic dissociation: when calcium chloride is introduced to water (h₂o), it undergoes a process called dissociation. The phase transitions of calcium chloride between various hydrates and solid–liquid phase transitions are common in many natural and industrial processes. recent studies have revealed some discrepancies in investigating the hydration and deliquescence of calcium chloride using different methods. Calcium chloride in water dissociates to provide calcium (ca 2 ) and chloride (cl ) ions. they are normal constituents of the body fluids and are dependent on various physiological mechanisms for maintenance of balance between intake and output.
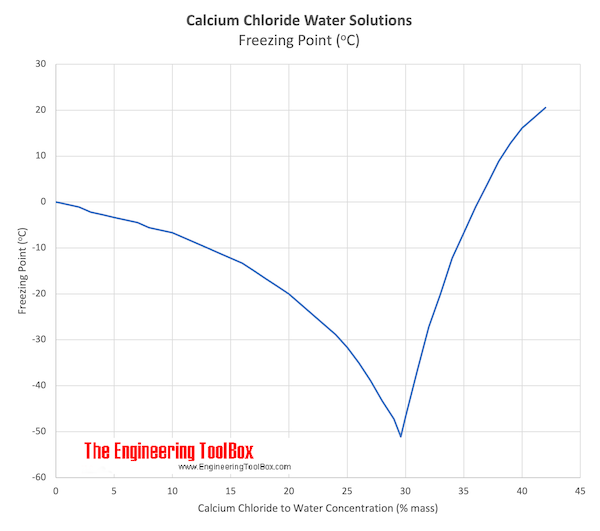
Calcium Chloride In Water The phase transitions of calcium chloride between various hydrates and solid–liquid phase transitions are common in many natural and industrial processes. recent studies have revealed some discrepancies in investigating the hydration and deliquescence of calcium chloride using different methods. Calcium chloride in water dissociates to provide calcium (ca 2 ) and chloride (cl ) ions. they are normal constituents of the body fluids and are dependent on various physiological mechanisms for maintenance of balance between intake and output. High purity calcium chloride plays a multifaceted role in water purification, contributing to the removal of impurities and the overall safety of treated water. Calcium chloride is a chemical compound made up of calcium and chlorine, known scientifically as cacl2. it appears as a white, powdery substance and is highly soluble in water. this compound is widely used to melt ice on roads, control dust, and act as a preservative in foods. The short answer to the question, "is calcium chloride in water safe", is a definitive yes. from a biological standpoint, calcium chloride dissociates into calcium ions and chloride ions upon entering the water. Calcium chloride is highly soluble, readily dissolving in water and separating into calcium ions and chloride ions. this compound is odorless, has a distinctive salty taste, and is hygroscopic, meaning it attracts and retains moisture from the air.
Comments are closed.