Buffer Solutions Explained Simply What Is A Buffer And How Does A Buffer Solution Work

Buffer Solutions Explained Simply What Is A Buffer And How Does A An aqueous solution that resists any change in ph by adding a small amount of acid or base is called a buffer solution. a buffer solution consists of a weak acid and its conjugate base or a weak base and its conjugate acid. A buffer is a solution that can resist ph change upon the addition of an acidic or basic components. it is able to neutralize small amounts of added acid or base, thus maintaining the ph of the solution relatively stable.

Buffer Solution Types Properties And Uses A buffer is a solution that resists changes in ph when small amounts of acid or base are added to it. it works by containing two chemical partners: a weak acid and its conjugate base (or a weak base and its conjugate acid). In chemistry, the definition of a buffer is a solution that can resist ph change upon the addition of an acid or a base. it consists of a solution of a weak acid and its conjugate base, or vice versa. a buffer is an extremely useful solution used in acid base chemistry. How do buffer solutions work? a buffer solution has to contain things which will remove any hydrogen ions or hydroxide ions that you might add to it otherwise the ph will change. A buffer solution is a solution that resists changes in hydrogen ion concentration when a modest amount of acid or base is added. in other words, such solutions are known to have reverse acidity and reverse basicity and to keep a reasonably steady ph value.
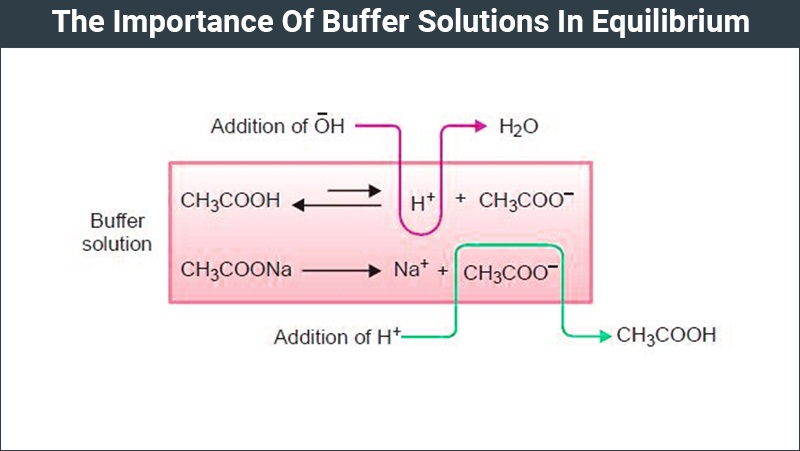
Buffer Solutions In Equilibrium Types Of Buffer Solution Chemistry How do buffer solutions work? a buffer solution has to contain things which will remove any hydrogen ions or hydroxide ions that you might add to it otherwise the ph will change. A buffer solution is a solution that resists changes in hydrogen ion concentration when a modest amount of acid or base is added. in other words, such solutions are known to have reverse acidity and reverse basicity and to keep a reasonably steady ph value. In this video i will give you a simple and easy to follow explanation of what exactly a buffer solution is, how a buffer solution is made and how a buffer solution works to keep. A buffer solution is a solution where the ph does not change significantly on dilution or if an acid or base is added at constant temperature. [1] its ph changes very little when a small amount of strong acid or base is added to it. A buffer is a solution containing acid and a proportionate amount of conjugate base capable of maintaining a stable ph when a small amount of additional acid or base is added to it. A buffer solution is an aqueous solution that resists changes in ph when small amounts of strong acid or base are added. it typically consists of a mixture of a weak acid and its conjugate base, or a weak base and its conjugate acid.
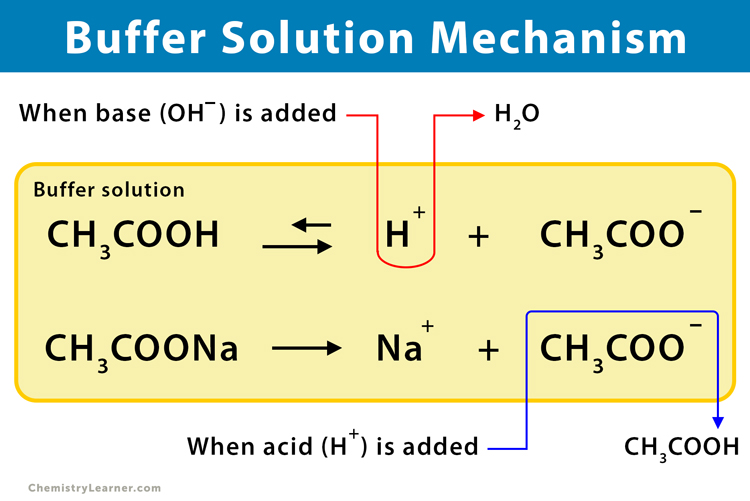
Buffer Solution Definition Examples And Preparation In this video i will give you a simple and easy to follow explanation of what exactly a buffer solution is, how a buffer solution is made and how a buffer solution works to keep. A buffer solution is a solution where the ph does not change significantly on dilution or if an acid or base is added at constant temperature. [1] its ph changes very little when a small amount of strong acid or base is added to it. A buffer is a solution containing acid and a proportionate amount of conjugate base capable of maintaining a stable ph when a small amount of additional acid or base is added to it. A buffer solution is an aqueous solution that resists changes in ph when small amounts of strong acid or base are added. it typically consists of a mixture of a weak acid and its conjugate base, or a weak base and its conjugate acid.
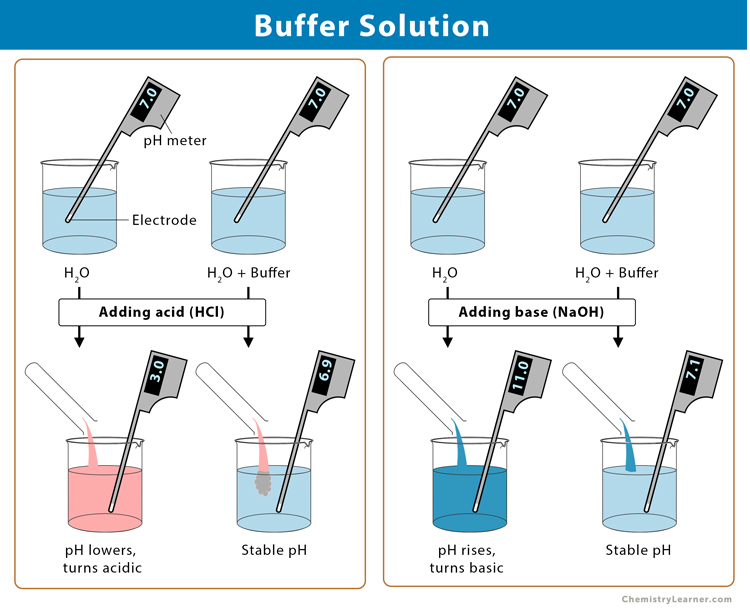
Buffer Solution Definition Examples And Preparation A buffer is a solution containing acid and a proportionate amount of conjugate base capable of maintaining a stable ph when a small amount of additional acid or base is added to it. A buffer solution is an aqueous solution that resists changes in ph when small amounts of strong acid or base are added. it typically consists of a mixture of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Comments are closed.