Bioscontrol Preservative Testing

Preservative Testing Isspllab Nowadays preservatives should not be only molecules with a formula protection activity but they need to be more: they have to meet the stability and micro testing requirements for high quality products to alleviate safety concerns. About press copyright contact us creators advertise developers terms privacy policy & safety how works test new features nfl sunday ticket © 2025 google llc.

Preservative Testing Services This article helps you to understand the basic considerations of preservative and antimicrobial efficacy testing, including determining a minimum inhibitory concentration (mic), giving you the background needed to select the best test methods for your food product evaluation. Standard operating procedure (sop) and guideline for preservative efficacy test in microbiology laboratory for checking of effectiveness of preservatives added in the drug formulations. The test shall be performed using either pour plate method or by membrane filtration, as appropriate and incubated at appropriate incubation by giving maximum incubation period, as given in the compendial chapter for antimicrobial effectiveness test. Italian active ingredient supplier roelmi hpc has launched bioscontrol evolve boreal, a preservative system made using upcycled pine tree by products.
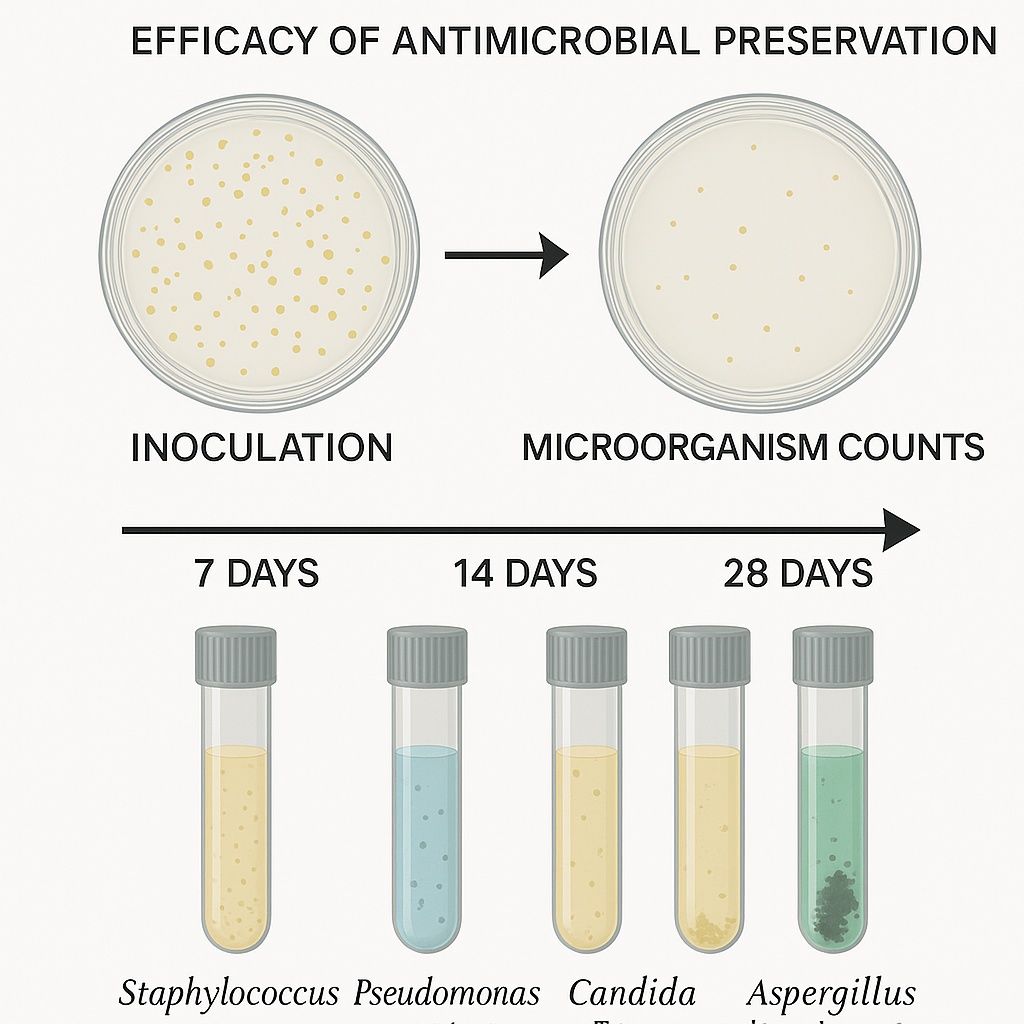
Preservative Effectiveness Testing Ph Eur 5 1 3 The test shall be performed using either pour plate method or by membrane filtration, as appropriate and incubated at appropriate incubation by giving maximum incubation period, as given in the compendial chapter for antimicrobial effectiveness test. Italian active ingredient supplier roelmi hpc has launched bioscontrol evolve boreal, a preservative system made using upcycled pine tree by products. Use the initial count of suspensions to calibrate the size of the inoculum to be used in the test. use the bacterial and candida albicans suspensions within 24 hours of harvest. fungal suspensions shall be refrigerated and used for up to 7 days. In contrast, samples without preservative agent showed no detectable ribosomal bands and a more pronounced degradation pattern. taken together, these findings suggest that the presence of guanidine edta may contribute to rna preservation during storage, although this interpretation should be considered in light of the inherent limitations of. To enable the identification of safer preservatives, substances from eight functional classes were assessed for antimicrobial efficacy and human and environmental hazards. This test should be performed on all aqueous based products that include ophthalmic, optic, nasal, oral, irrigation, and dialysis fluids. testing performed by nelson labs meets the criteria set forth by the usp general chapter 51 on antimicrobial effectiveness testing.

Preservative Efficacy Testing Use the initial count of suspensions to calibrate the size of the inoculum to be used in the test. use the bacterial and candida albicans suspensions within 24 hours of harvest. fungal suspensions shall be refrigerated and used for up to 7 days. In contrast, samples without preservative agent showed no detectable ribosomal bands and a more pronounced degradation pattern. taken together, these findings suggest that the presence of guanidine edta may contribute to rna preservation during storage, although this interpretation should be considered in light of the inherent limitations of. To enable the identification of safer preservatives, substances from eight functional classes were assessed for antimicrobial efficacy and human and environmental hazards. This test should be performed on all aqueous based products that include ophthalmic, optic, nasal, oral, irrigation, and dialysis fluids. testing performed by nelson labs meets the criteria set forth by the usp general chapter 51 on antimicrobial effectiveness testing.

Preclinical Preservative Testing Services For Safe Cosmetic Formulations To enable the identification of safer preservatives, substances from eight functional classes were assessed for antimicrobial efficacy and human and environmental hazards. This test should be performed on all aqueous based products that include ophthalmic, optic, nasal, oral, irrigation, and dialysis fluids. testing performed by nelson labs meets the criteria set forth by the usp general chapter 51 on antimicrobial effectiveness testing.
Antimicrobial Preservative Efficacy Testing Nelson Labas
Comments are closed.