Basic Thermodynamic Systems

Premium Ai Image Fundamentals Of Thermodynamic Systems We will start these lectures by considering closed systems, i.e. systems with n = cst, for simplicity and come back to systems where the number of particles can vary after we have discussed the fundamental laws of thermodynamics. 13. the basic principles of thermodynamics # thermodynamics is the study of heat, its connection to energy, and the static (equilibrium) and dynamic properties of systems that have a thermal component.

Thermodynamic Terms And Basic Concept Thermodynamic laws describe the fundamental principles that govern energy, temperature, entropy, and their behavior in thermodynamic systems. these laws explain how energy is transferred and transformed under different conditions. Thermodynamics is the study of the relations between heat, work, temperature, and energy. the laws of thermodynamics describe how the energy in a system changes and whether the system can perform useful work on its surroundings. A thermodynamic system includes anything whose thermodynamic properties are of interest. it is embedded in its surroundings or environment; it can exchange heat with, and do work on, its environment …. •explain the basic concepts of thermodynamics such as system, state, equilibrium, process, and cycle. •discuss properties of a system and define density, specific gravity, and specific weight.
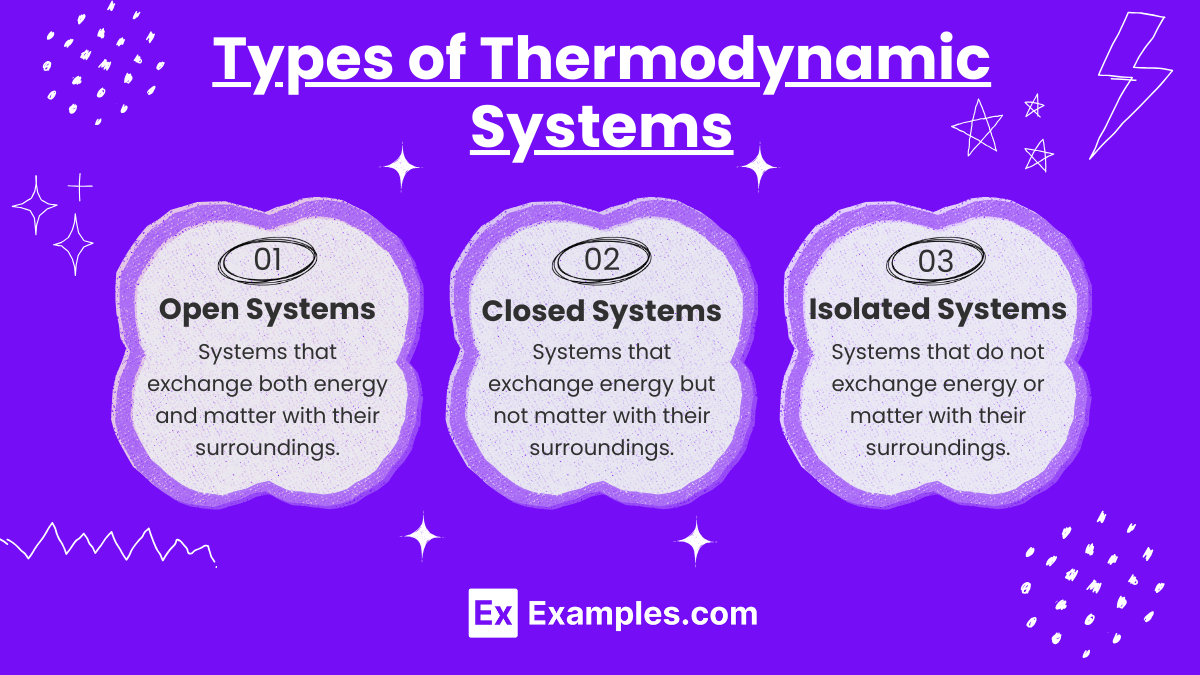
Unit 2 1 Thermodynamic Systems Notes Practice Questions Ap A thermodynamic system includes anything whose thermodynamic properties are of interest. it is embedded in its surroundings or environment; it can exchange heat with, and do work on, its environment …. •explain the basic concepts of thermodynamics such as system, state, equilibrium, process, and cycle. •discuss properties of a system and define density, specific gravity, and specific weight. Goal of the lecture: to study the fundamental principles of thermodynamics, including the classification of systems, their properties, and parameters used to describe their state. Thermodynamics is based on four fundamental laws — the zeroth, first, second, and third laws. each law describes a key principle of heat and energy. if two systems are each in thermal equilibrium with a third system, they are in equilibrium with each other. The thermodynamic state of a system is defined by specifying a set of measurable properties sufficient so that all remaining properties are determined. examples of properties: pressure, temperature, density, internal energy, enthalpy, and entropy. Lecture notes covering basic thermodynamics concepts, laws, systems, properties, energy, work, and heat. ideal for mechanical engineering students.
Comments are closed.