Atomic Theory Pptx Third Quarter Lesson Pptx

Atomic Theory Pptx Third Quarter Lesson Pptx Subsequent discoveries by scientists like thomson, rutherford, and bohr expanded the understanding of atomic structure, revealing subatomic particles and the arrangement of electrons in orbits around a dense nucleus. download as a pptx, pdf or view online for free. Lesson 2: the atomic theory presentation: drive.google file d 1i5yprhjmytocj byqmwnwfpl0pevg3x view?usp=drive link.
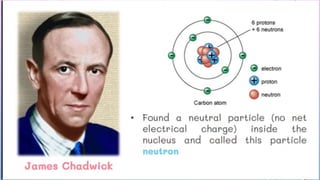
Atomic Theory Pptx Third Quarter Lesson Pptx G8 science q3 ppt atomic structure free download as powerpoint presentation (.ppt .pptx), pdf file (.pdf), text file (.txt) or view presentation slides online. the document discusses the atomic structure, including the history of atomic theory from democritus to rutherford's model of the nucleus. Atomic structure atoms are made up of three subatomic particles: protons, neutrons, and electrons. protons and neutrons are found in the nucleus, while electrons orbit around the nucleus in shells. the number of protons in an atom determines its atomic number and the element it represents. 1) models of the atom have evolved over time as new evidence and theories emerged. early models included thomson's "plum pudding" model and rutherford's nuclear model. 2) bohr's model improved on earlier work by proposing that electrons orbit the nucleus in fixed, quantized energy levels. Atomic theory.pptx third quarter lesson. this document discusses the development of atomic theory over time. it describes early greek philosopher democritus' idea that matter is made of indivisible particles called atoms.

Atomic Theory Pptx Third Quarter Lesson Pptx 1) models of the atom have evolved over time as new evidence and theories emerged. early models included thomson's "plum pudding" model and rutherford's nuclear model. 2) bohr's model improved on earlier work by proposing that electrons orbit the nucleus in fixed, quantized energy levels. Atomic theory.pptx third quarter lesson. this document discusses the development of atomic theory over time. it describes early greek philosopher democritus' idea that matter is made of indivisible particles called atoms. The document outlines the history and development of atomic theory, highlighting contributions from key scientists like john dalton, j.j. thomson, ernest rutherford, and james chadwick. Lesson 2 describes j.j. thomson's plum pudding model of the atom and ernest rutherford's nuclear model. it includes an activity where students answer questions about the models. lesson 3 will cover the number of subatomic particles in different atomic structures and includes a closing activity. History of the periodic table trends in a periodic table.pptx. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. modern atomic theory. all matter is composed of atoms. atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. however, these changes can occur in nuclear reactions!.

Atomic Theory Pptx Third Quarter Lesson Pptx The document outlines the history and development of atomic theory, highlighting contributions from key scientists like john dalton, j.j. thomson, ernest rutherford, and james chadwick. Lesson 2 describes j.j. thomson's plum pudding model of the atom and ernest rutherford's nuclear model. it includes an activity where students answer questions about the models. lesson 3 will cover the number of subatomic particles in different atomic structures and includes a closing activity. History of the periodic table trends in a periodic table.pptx. Students know the nucleus of the atom is much smaller than the atom yet contains most of its mass. modern atomic theory. all matter is composed of atoms. atoms cannot be subdivided, created, or destroyed in ordinary chemical reactions. however, these changes can occur in nuclear reactions!.
Comments are closed.