Answered %d0%be%d0%bd H2so4 Nacl Bartleby
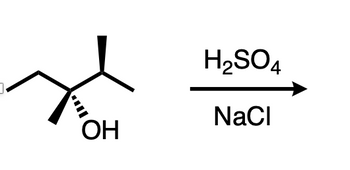
Answered он H2so4 Nacl Bartleby Enter an equation of a chemical reaction and click 'balance'. the answer will appear below. always use the upper case for the first character in the element name and the lower case for the second character. examples: fe, au, co, br, c, o, n, f. compare: co cobalt and co carbon monoxide. Balance the reaction of h2so4 nacl = na2so4 hcl using this chemical equation balancer!.

Answered Data Related To Aqueous Solutions Of Bartleby Solved and balanced chemical equation nacl h2so4 → nahso4 hcl with completed products. application for completing products and balancing equations. Hint: the reaction between sodium chloride and sulphuric acid gives hydrogen chloride. the hydrogen chloride is gas so it escapes from the medium. concentrated sulphuric acid works as a dehydrating agent, so it discards water molecules from the compound and thus dehydrates the compound. Let's balance this equation using the inspection method. for each element, we check if the number of atoms is balanced on both sides of the equation. na is not balanced: 1 atom in reagents and 2 atoms in products. cl is not balanced: 2 atoms in reagents and 1 atom in products. h is balanced: 2 atoms in reagents and 2 atoms in products. In this chemical reaction, 1 moles of sodium chloride (nacl) and 1 moles of sulfuric acid (h 2 so 4) react to form 1 moles of sodium bisulfate (nahso 4) and 1 moles of hydrogen chloride (hcl). the given chemical equation is already balanced. for learning purposes, the algebraic method is shown below to verify the coefficients.

Solved In An Experiment Nacl Is Reacted With H2so4 And Evolves Hcl Gas Let's balance this equation using the inspection method. for each element, we check if the number of atoms is balanced on both sides of the equation. na is not balanced: 1 atom in reagents and 2 atoms in products. cl is not balanced: 2 atoms in reagents and 1 atom in products. h is balanced: 2 atoms in reagents and 2 atoms in products. In this chemical reaction, 1 moles of sodium chloride (nacl) and 1 moles of sulfuric acid (h 2 so 4) react to form 1 moles of sodium bisulfate (nahso 4) and 1 moles of hydrogen chloride (hcl). the given chemical equation is already balanced. for learning purposes, the algebraic method is shown below to verify the coefficients. The balanced equation for the reaction of sodium chloride, nacl, with sulphuric acid, h2so4, is as follows: 2nacl (s) h2so4 (aq) → na2so4 (aq) 2hcl (g) during a reaction, 1.5 g of an impure sample of sodium chloride reacts with 100 cm³ sulphuric acid of concentration 0,1 mol·dm⁻³ at room temperature. **6.2.1** define the term. 2nacl h2so4 → na2so4 2hcl. the coefficients show the number of particles (atoms or molecules), and the indices show the number of atoms that make up the molecule. new substances are formed as a result of the rearrangement of the original atoms. Solved and balanced chemical equation 2 nacl h2so4 → 2 hcl na2so4 with completed products. application for completing products and balancing equations. In this video we'll balance the equation nacl h2so4 = na2so4 hcl and provide the correct coefficients for each compound.

Answered 1 The Table Below Contains Partial Bartleby The balanced equation for the reaction of sodium chloride, nacl, with sulphuric acid, h2so4, is as follows: 2nacl (s) h2so4 (aq) → na2so4 (aq) 2hcl (g) during a reaction, 1.5 g of an impure sample of sodium chloride reacts with 100 cm³ sulphuric acid of concentration 0,1 mol·dm⁻³ at room temperature. **6.2.1** define the term. 2nacl h2so4 → na2so4 2hcl. the coefficients show the number of particles (atoms or molecules), and the indices show the number of atoms that make up the molecule. new substances are formed as a result of the rearrangement of the original atoms. Solved and balanced chemical equation 2 nacl h2so4 → 2 hcl na2so4 with completed products. application for completing products and balancing equations. In this video we'll balance the equation nacl h2so4 = na2so4 hcl and provide the correct coefficients for each compound.
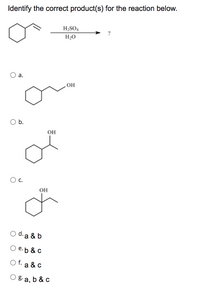
Answered Dentify The Correct Product S For The Reaction Below H2so4 Solved and balanced chemical equation 2 nacl h2so4 → 2 hcl na2so4 with completed products. application for completing products and balancing equations. In this video we'll balance the equation nacl h2so4 = na2so4 hcl and provide the correct coefficients for each compound.
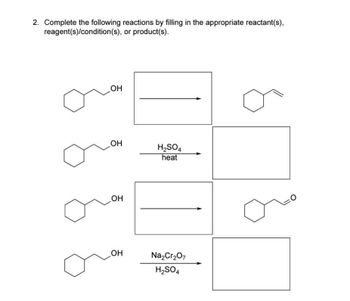
Answered 2 Complete The Following Reactions By Filling In The
Comments are closed.