Acidic Hydrogens Mpg
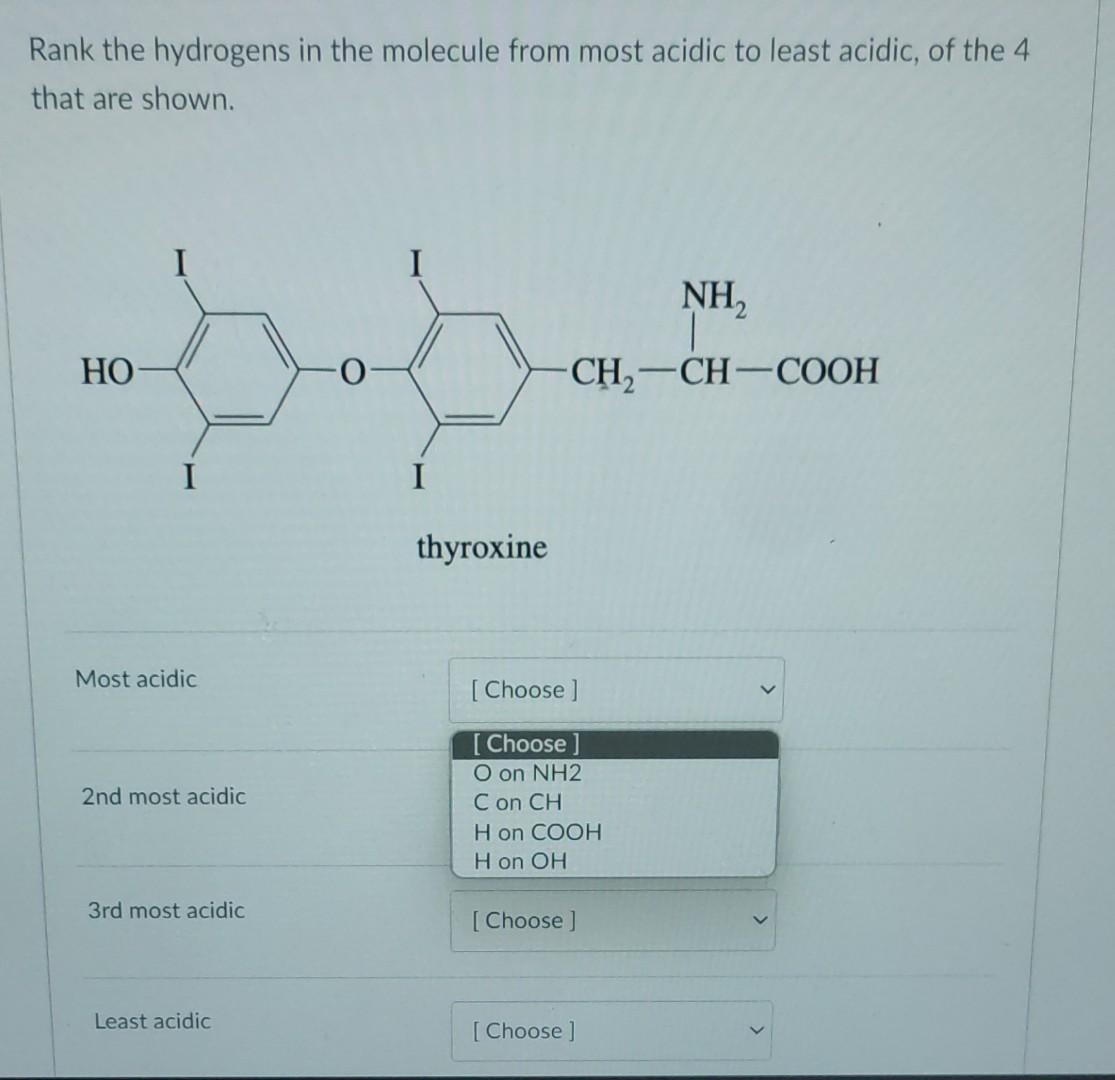
Solved Rank The Hydrogens In The Molecule From Most Acidic Chegg When given a molecule with multiple hydrogen atoms, how does one tell which is the most acidic hydrogen atom?. There are five key factors that influence acidity in organic chemistry; the charge, the atom, resonance, inductive effects, and the orbitals.
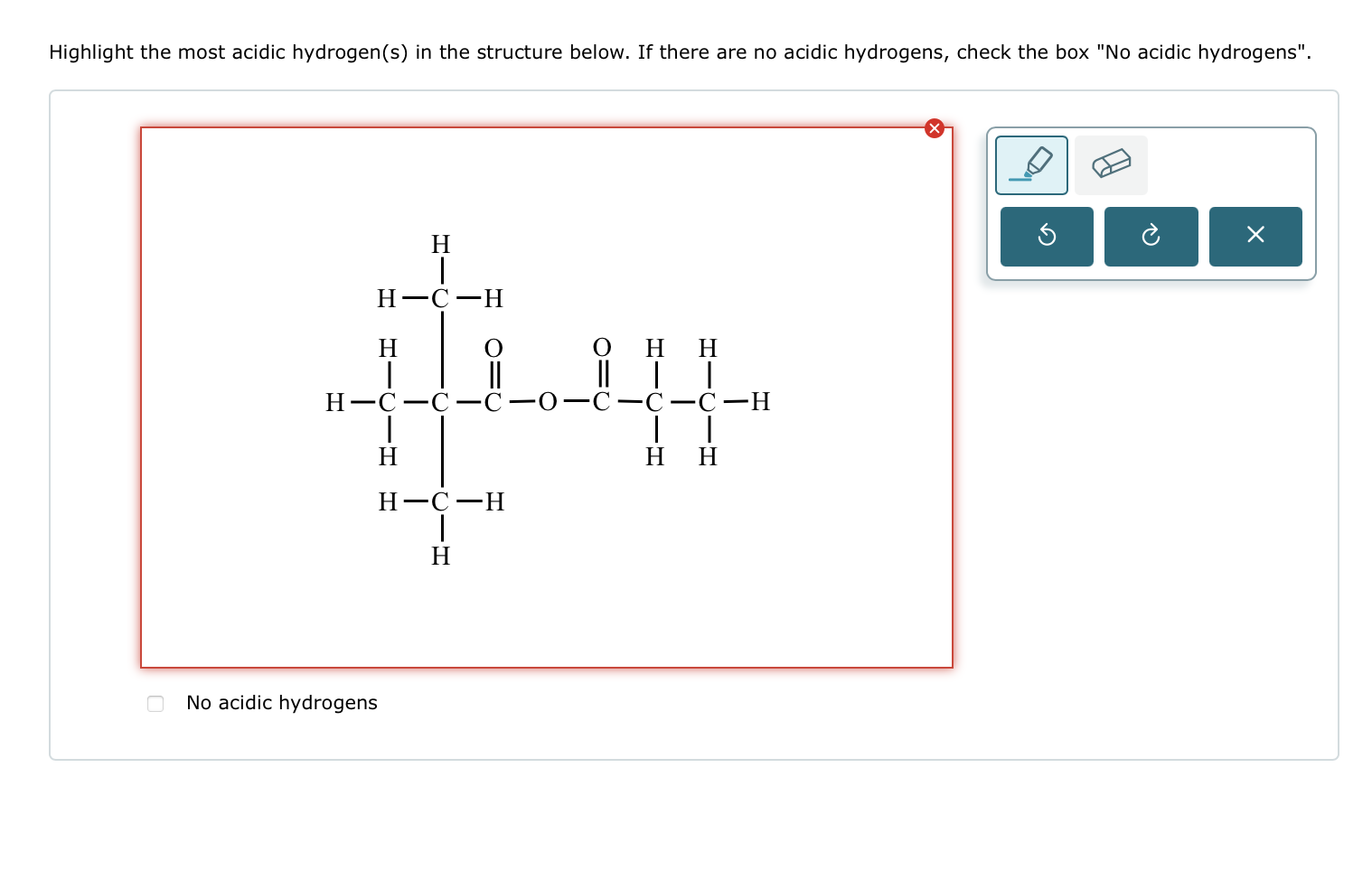
Solved Highlight The Most Acidic Hydrogen S ï In The Chegg Understanding what makes a hydrogen acidic is necessary to predict how a substance will react in chemical and biological environments. an acidic hydrogen is a proton that a molecule can easily lose or donate to a base. The presence of these overlapping p orbitals gives α hydrogens (hydrogens on carbons adjacent to carbonyls) special properties. in particular, α hydrogens are weakly acidic because the conjugate base, called an enolate, is stabilized though conjugation with the π orbitals of the carbonyl. Many compounds, such as acetic acid (ch 3 cooh), contain many hydrogens, not all of which are acidic. in this section, we will go over how to identify the most acidic hydrogen or most basic atom in a molecule based on its structure. Hydrogens on carbon next to a carbonyl group are acidic. in general, a β dicarbonyl compound is most acidic, a ketone or aldehyde is next most acidic, and a carboxylic acid derivative is least acidic.
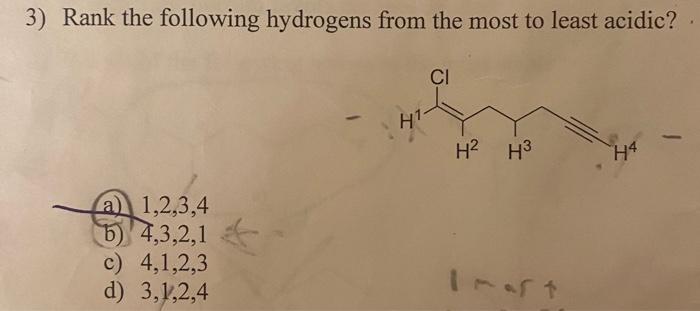
Solved Rank The Following Hydrogens From The Most Acidic To Chegg Many compounds, such as acetic acid (ch 3 cooh), contain many hydrogens, not all of which are acidic. in this section, we will go over how to identify the most acidic hydrogen or most basic atom in a molecule based on its structure. Hydrogens on carbon next to a carbonyl group are acidic. in general, a β dicarbonyl compound is most acidic, a ketone or aldehyde is next most acidic, and a carboxylic acid derivative is least acidic. This document discusses the acidity of hydrogens bonded to carbon atoms, particularly focusing on the differences in acidity between sp3 carbons, aldehydes, ketones, and esters. When studying organic chemistry, understanding the relative acidity of different functional groups is crucial for predicting reaction mechanisms and product outcomes. this foundational concept often hinges on identifying what constitutes an acidic hydrogen within various organic compounds. We can rationalise the trend by comparing the two structures, the difference simply being the alkyl group versus the hydrogen. since alkyl groups are weakly electron donating, they tend to destabilise anions (you should recall that they stabilise carbocations). Acidic solutions have excess h which will react with the oh– in the basic salt, causing the equation to shift in the forward direction, increasing solubility by using more salt.
Comments are closed.