Acidic Basic And Neutral Salts Compounds

Acidic Basic And Neutral Salts Ionic Compounds Video Summary And Learn how salt hydrolysis affects solution ph. explore acidic, basic, and neutral salts with clear explanations, examples, and practice problems. Sodium chloride salt (nacl) is crystallized from a strong acid hydrochloric acid (hcl) and a strong base sodium hydroxide (naoh). since sodium chloride is formed from a strong acid and a strong base, thus, an aqueous solution of sodium chloride is neutral.
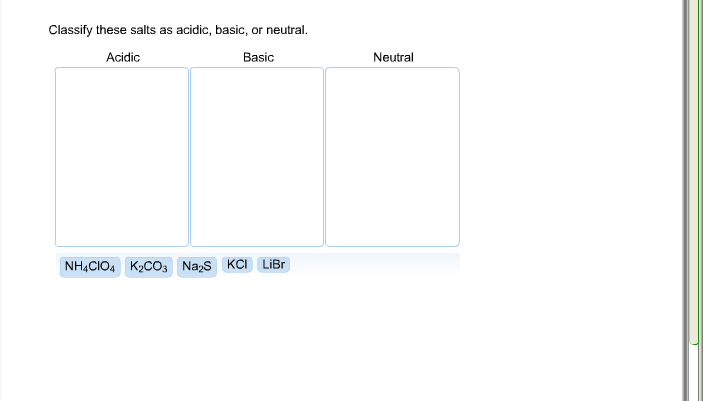
Solved Classify These Salts As Acidic Basic Or Neutral Chegg Identify a salt solution as acidic, basic, or neutral. a salt is an ionic compound that is formed when an acid and a base neutralize each other. while it may seem that salt solutions are always neutral, they can frequently be either acidic or basic. Learn about acidic, basic, and neutral salts, their formation through acid base reactions, and how they affect solution ph in chemistry. What are acidic, basic and neutral salts? this video explores the acidic, basic and neutral nature of various salts. you will learn how to identify these different types of salts. A salt of strong acid and a strong base will be a neutral salt. a salt of weak acid and weak base can be acidic, basic, or neutral depending upon the strengths of weak acid and weak base.
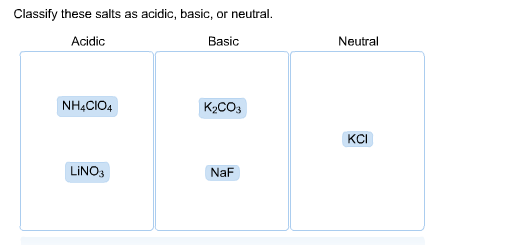
Solved Classify These Salts As Acidic Basic Or Neutral Chegg What are acidic, basic and neutral salts? this video explores the acidic, basic and neutral nature of various salts. you will learn how to identify these different types of salts. A salt of strong acid and a strong base will be a neutral salt. a salt of weak acid and weak base can be acidic, basic, or neutral depending upon the strengths of weak acid and weak base. This chemistry video explains how to determine if an ionic compound or a salt is acidic, basic, or neutral. acidic ions include the ammonium ion and metal cations such as al3 and fe3 . Depending on the composition of the salt (the ions which it is made up of) the solution will be either acidic or basic. soluble salts that contain anions derived from weak acids form solutions that are basic. Salts are ionic compounds resulting from a neutralisation reaction between an acid and a base. depending on the respective strengths of the reactants, the types of salts in chemistry can be classified as acidic, basic, normal (neutral), or double. 4.the salt of a weak acid and a weak base hydrolyzes to produce a slightly acidic, neutral, or slightly basic solution, depending on the relative weaknesses of the acid and base.
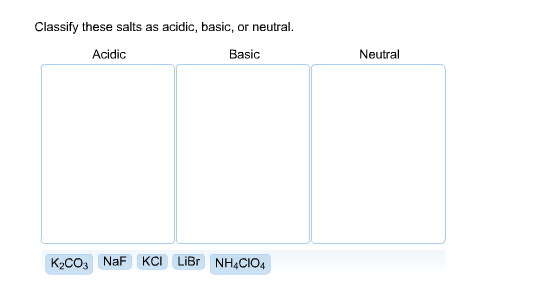
Solved Classify These Salts As Acidic Basic Or Neutral Chegg This chemistry video explains how to determine if an ionic compound or a salt is acidic, basic, or neutral. acidic ions include the ammonium ion and metal cations such as al3 and fe3 . Depending on the composition of the salt (the ions which it is made up of) the solution will be either acidic or basic. soluble salts that contain anions derived from weak acids form solutions that are basic. Salts are ionic compounds resulting from a neutralisation reaction between an acid and a base. depending on the respective strengths of the reactants, the types of salts in chemistry can be classified as acidic, basic, normal (neutral), or double. 4.the salt of a weak acid and a weak base hydrolyzes to produce a slightly acidic, neutral, or slightly basic solution, depending on the relative weaknesses of the acid and base.
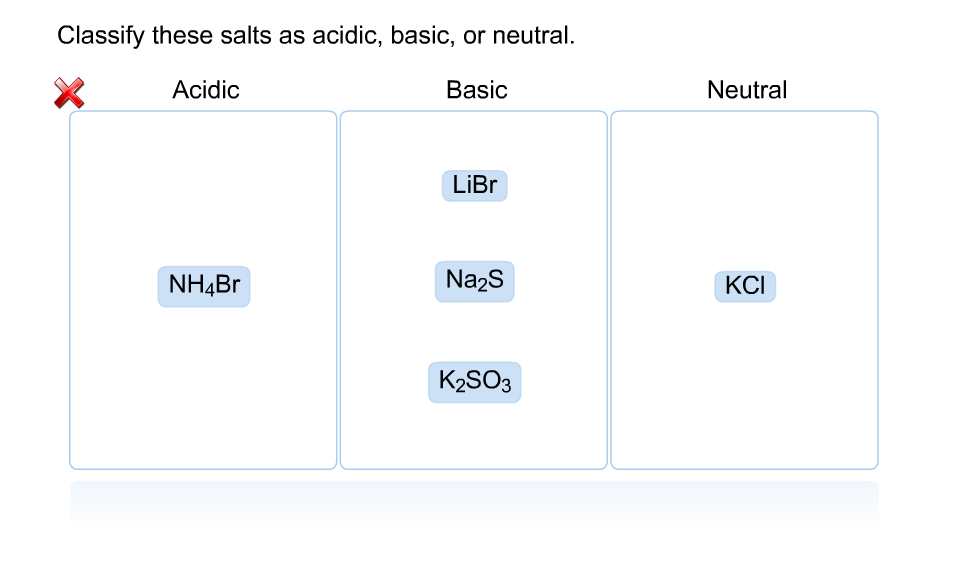
Solved Classify These Salts As Acidic Basic Or Neutral Chegg Salts are ionic compounds resulting from a neutralisation reaction between an acid and a base. depending on the respective strengths of the reactants, the types of salts in chemistry can be classified as acidic, basic, normal (neutral), or double. 4.the salt of a weak acid and a weak base hydrolyzes to produce a slightly acidic, neutral, or slightly basic solution, depending on the relative weaknesses of the acid and base.
Comments are closed.