Abbott Advances Two Pulsed Field Ablation Studies For Afib Treatment

Abbott Advances Two Pulsed Field Ablation Studies For Afib Treatment Together, these two studies represent significant advances for the future of two abbott pfa catheters, which are being developed as important tools for physicians treating people with abnormal heart rhythms like atrial fibrillation (afib). Abbott has reached two milestones in a pair of trials evaluating the company’s pulsed field ablation (pfa) technology for the treatment of abnormal heart rhythms. the first milestone came in the form of enrollment in the volt af study, which abbott said happened ahead of time.

Pulsed Field Afib Ablation Hits Endpoints In Pivotal Trial Gcg Global Together, these two studies represent significant advances for the future of two abbott pfa catheters, which are being developed as important tools for physicians treating people with. Discover the clinical evidence and trial data on abbott's volt™ pulse field ablation (pfa) system for treating atrial fibrillation. Together, these two studies represent significant advances for the future of two abbott pfa catheters, which are being developed as important tools for physicians treating people with abnormal heart rhythms like af, a company press release notes. Abbott (nyse: abt) today announced new clinical data from two late breaking presentations at af symposium in boston (february 5 7, 2026) that demonstrate the strong safety and efficacy of the company’s minimally invasive therapies to treat people with atrial fibrillation (afib).
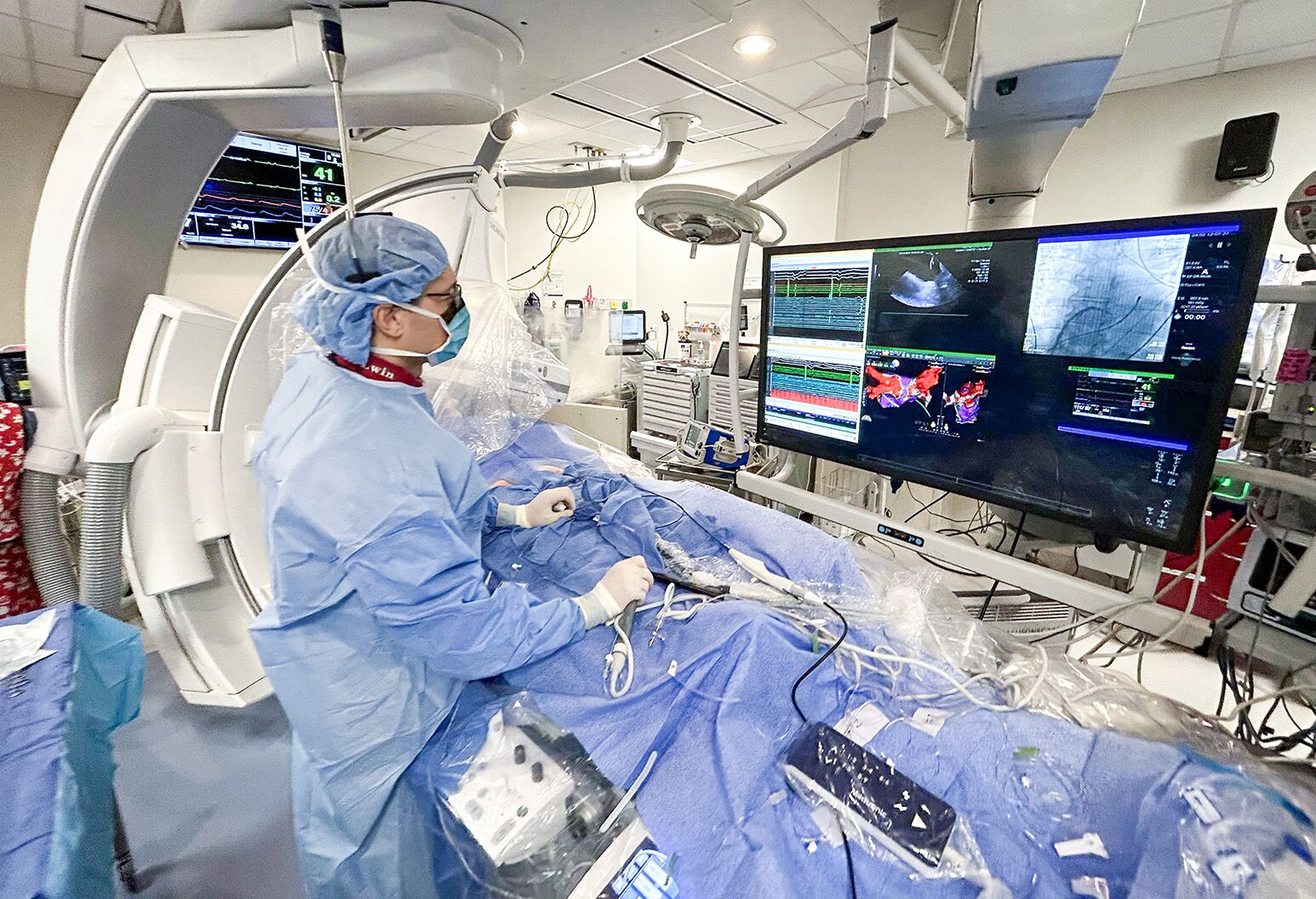
Pulsed Field Ablation Helps Conquer Afib Northwell Health Together, these two studies represent significant advances for the future of two abbott pfa catheters, which are being developed as important tools for physicians treating people with abnormal heart rhythms like af, a company press release notes. Abbott (nyse: abt) today announced new clinical data from two late breaking presentations at af symposium in boston (february 5 7, 2026) that demonstrate the strong safety and efficacy of the company’s minimally invasive therapies to treat people with atrial fibrillation (afib). Abbott (nyse: abt) today announced new clinical data supporting its minimally invasive therapies for treating afib. The results include 12 month findings that reinforce the long term safety and performance of abbott's volt™ pulsed field ablation (pfa) system, which were simultaneously published in jacc: clinical electrophysiology. Abbott designed its volt ce mark study to assess the impact of the volt pfa system in two different patient groups – people battling paroxysmal atrial fibrillation (paf) episodes that come and go as well as persistent afib (persaf) episodes that last longer than seven days.

Pulsed Field Ablation Safe For Afib Patients Large New Study Confirms Abbott (nyse: abt) today announced new clinical data supporting its minimally invasive therapies for treating afib. The results include 12 month findings that reinforce the long term safety and performance of abbott's volt™ pulsed field ablation (pfa) system, which were simultaneously published in jacc: clinical electrophysiology. Abbott designed its volt ce mark study to assess the impact of the volt pfa system in two different patient groups – people battling paroxysmal atrial fibrillation (paf) episodes that come and go as well as persistent afib (persaf) episodes that last longer than seven days.
Comments are closed.