20 3 1 20 3 2 E2 Mechanism

E2 Mechanism Pdf The key difference between the s n 2 and e2 reactions is that the nucleophile in the s n 2 mechanism attacks the carbon connected to the leaving group (ɑ carbon) while in e2, the base attacks one of the β hydrogens. E2 mechanism the e2 mechanism takes place in a single concerted step. the rate at which this mechanism occurs follows second order kinetics, and depends on the concentration of both the base and alkyl halide. the base removes a hydrogen from a carbon adjacent to the carbon with the leaving group.
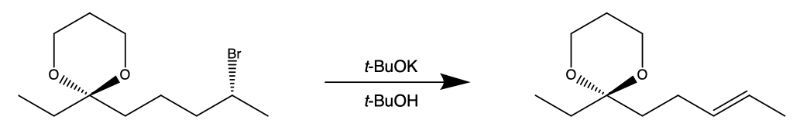
Exam Prep For Organic Chemistry Channels For Pearson Since it’s an elimination reaction, and the rate law is “bimolecular”, we call this mechanism the e2. in the next post, we’ll directly compare the e1 and e2 reactions. The mechanism of the e2 reaction is a single step concerted process with one transition state. first, the base removes the proton from the beta carbon, and the electron pair in the beta carbon forms a pi bond with the alpha carbon. In this section, we will discuss some important elimination mechanisms like e2, e1, and e1cb type. an e2 elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in a one step concerted mechanism. The e2 reaction process is a bimolecular elimination mechanism that removes beta hydrogen and a leaving group from a molecule (substrate) simultaneously to generate a double bond. the removal often occurs in the presence of a strong base that removes the proton.
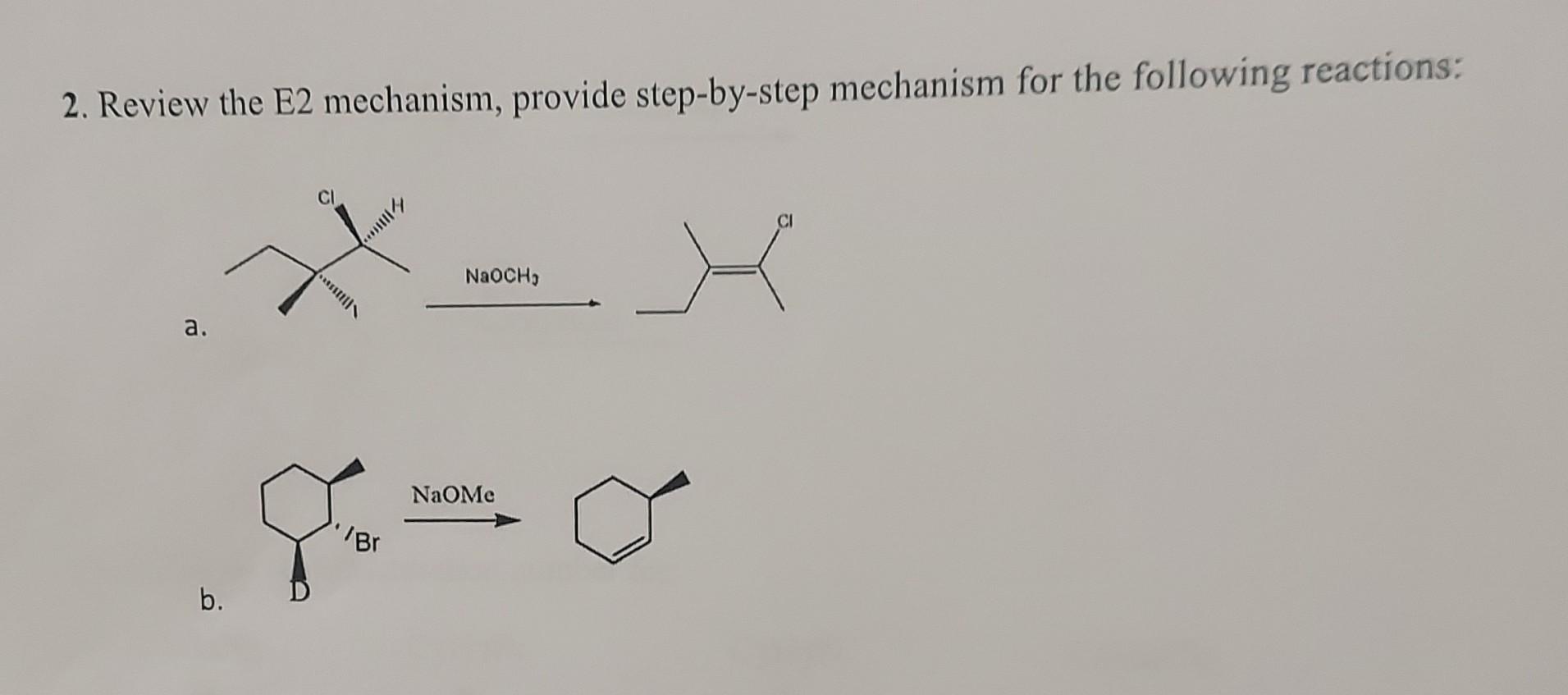
Solved 2 Review The E2 Mechanism Provide Step By Step Chegg In this section, we will discuss some important elimination mechanisms like e2, e1, and e1cb type. an e2 elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in a one step concerted mechanism. The e2 reaction process is a bimolecular elimination mechanism that removes beta hydrogen and a leaving group from a molecule (substrate) simultaneously to generate a double bond. the removal often occurs in the presence of a strong base that removes the proton. The most common mechanism is e2 elimination, which is a bimolecular reaction promoted by a strong base. it follows second order kinetics and has a single transition state. The document discusses the e2 elimination reaction mechanism. the e2 reaction involves the one step removal of two substituents from a molecule to form an alkene. E 2 reaction is also known as elimination bimolecular reaction. this reaction occurs when an alkyl halide is treated with a strong base such as hydroxide ion (oh ) and forms a carbon carbon double bond. 20.3.1 describe, using equations, the elimination of hbr from bromoalkanes.20.3.2 describe and explain the mechanism for the elimination of hbr from bromoalk.
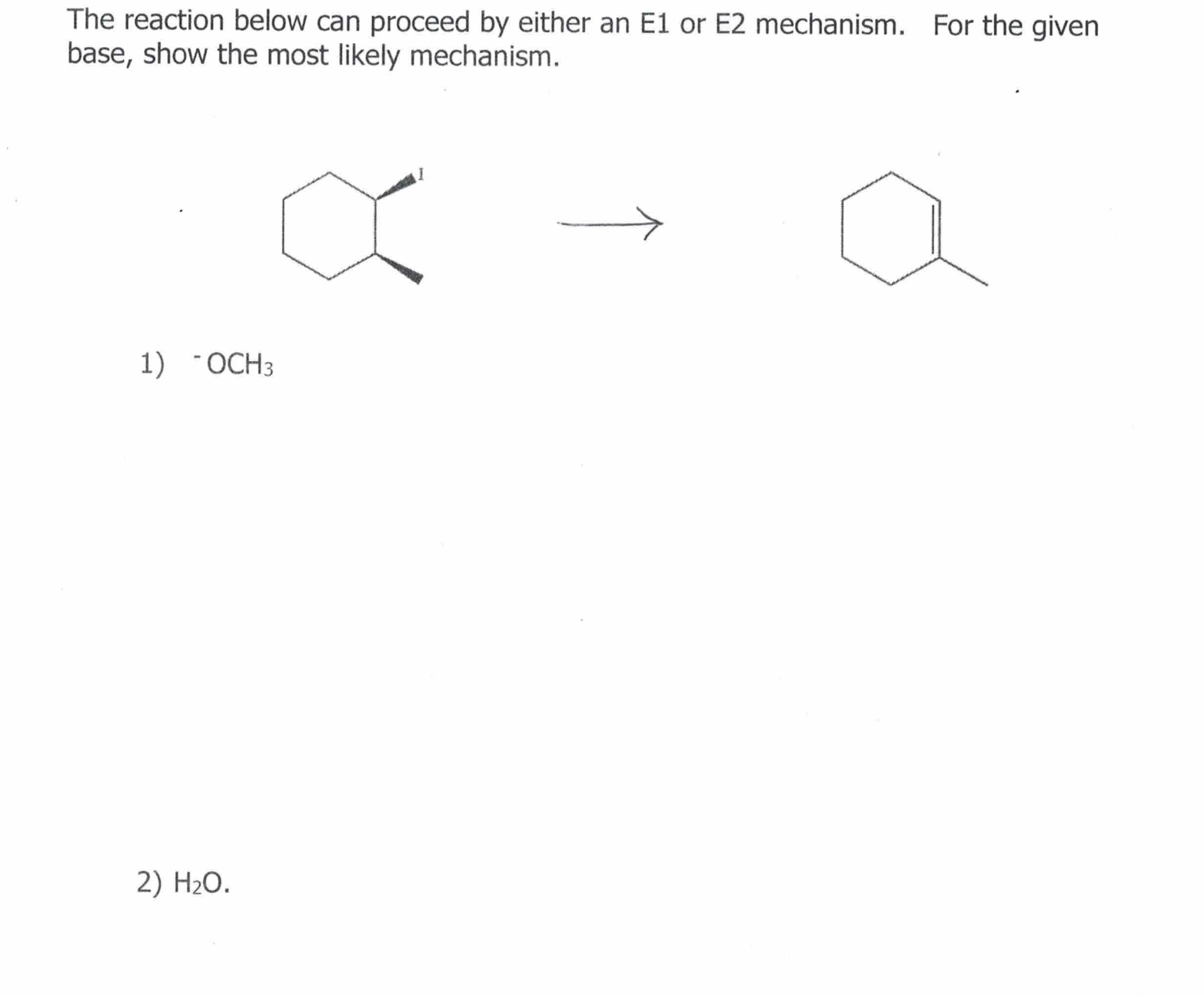
Solved The Reaction Below Can Proceed By Either An E1 Or E2 Chegg The most common mechanism is e2 elimination, which is a bimolecular reaction promoted by a strong base. it follows second order kinetics and has a single transition state. The document discusses the e2 elimination reaction mechanism. the e2 reaction involves the one step removal of two substituents from a molecule to form an alkene. E 2 reaction is also known as elimination bimolecular reaction. this reaction occurs when an alkyl halide is treated with a strong base such as hydroxide ion (oh ) and forms a carbon carbon double bond. 20.3.1 describe, using equations, the elimination of hbr from bromoalkanes.20.3.2 describe and explain the mechanism for the elimination of hbr from bromoalk.
Comments are closed.