2 Atomic Structure
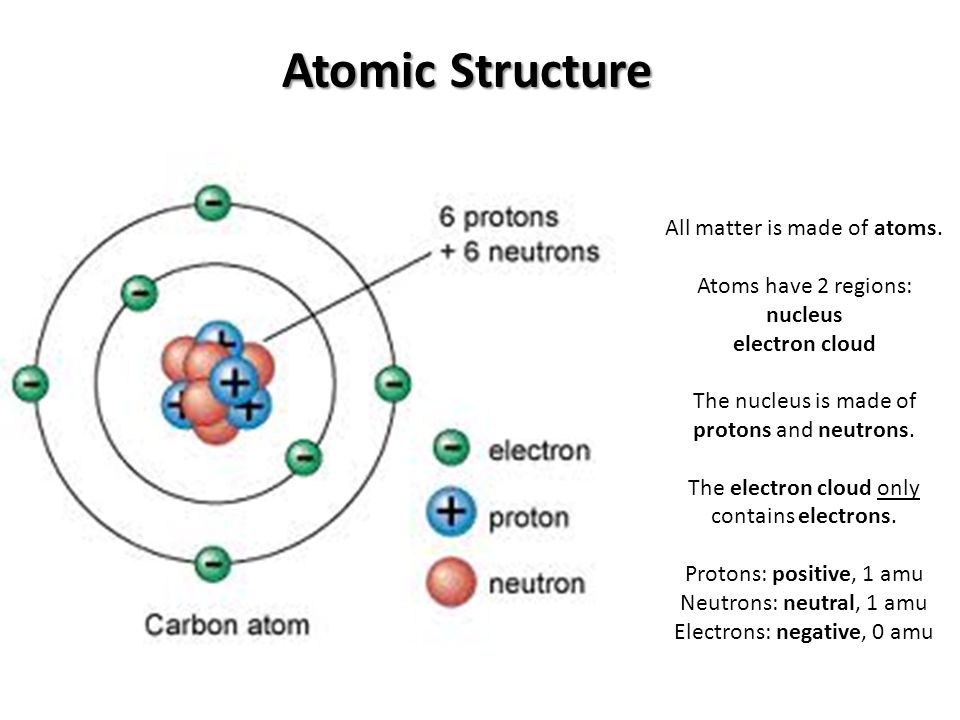
Atomic Structure Of Matter Science Online Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. atoms themselves are composed of protons, neutrons, and electrons. Atomic structure underpins the behaviour of elements, their interactions, and the formation of molecules. these are fundamental to chemistry, physics, and many applied sciences. this resource explains the structure of atoms, atomic numbers and mass numbers, and isotopes and atomic masses.

Atom Structure With Protons Neutrons Electrons Stock Illustration Learn about atomic structure for your igcse chemistry exam. find information on subatomic particles and understand how the atomic model has evolved. Atomic structure is the focus of this session, including a review of the famous backscattering experiment of rutherford. An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. What are the key learning points about atomic structure? there have been a number of ideas over time about the structure of atoms close atomthe smallest particle of an element.
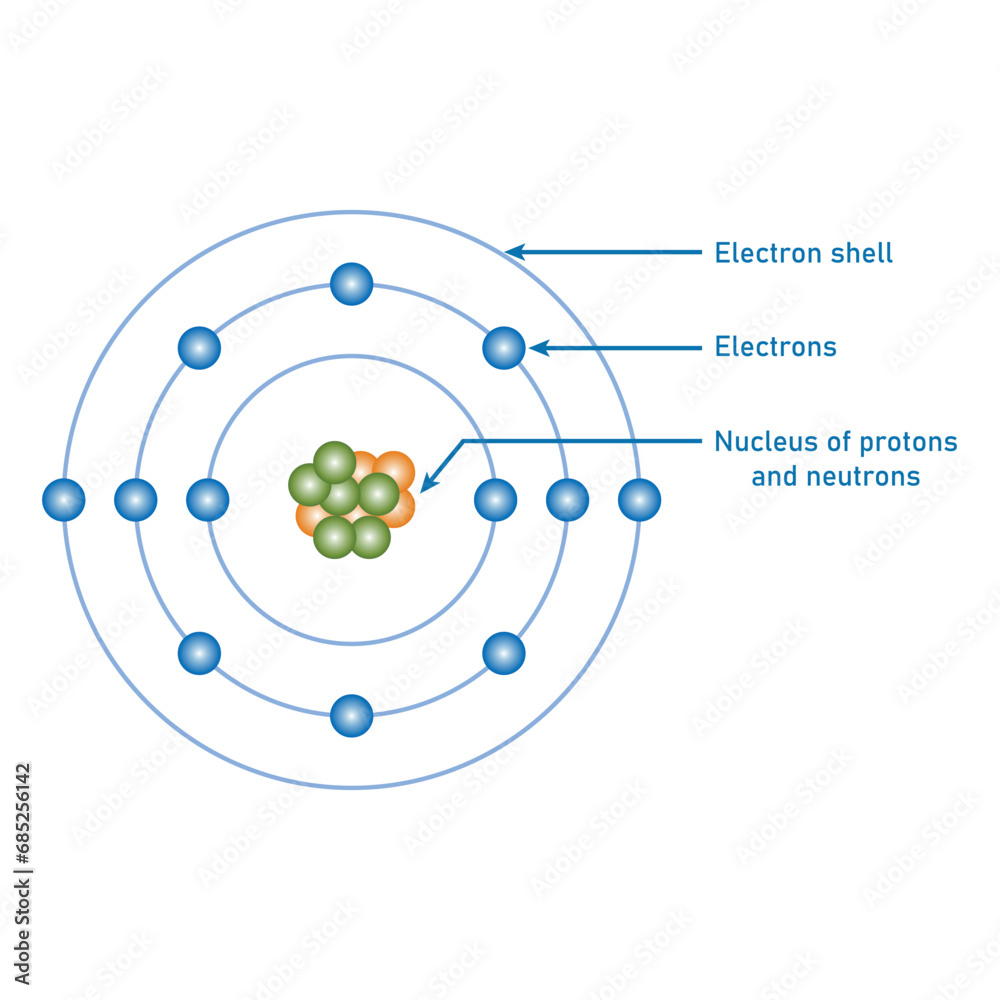
Atom Structure Model Diagram Bohr Atomic Model Of Atom Electrons An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. What are the key learning points about atomic structure? there have been a number of ideas over time about the structure of atoms close atomthe smallest particle of an element. Chemical elements are made up of only one type of atom—gold contains only gold atoms, and neon contains only neon atoms and they are ranked in order of their atomic number (the total number of protons in its nucleus) in a chart called the periodic table. If one is oriented along the x axis, called 2 p x, the second is along the y axis, called 2 p y, and the third is along the z axis, called 2 p z, as illustrated in figure 1.2.3. The development of modern atomic theory revealed much about the inner structure of atoms. it was learned that an atom contains a very small nucleus composed of positively charged protons and uncharged neutrons, surrounded by a much larger volume of space containing negatively charged electrons. These levels may be designated k l m n shells (or 1 2 3 4 orbits). the first level contains only 2 electrons. the second level can hold 6 electrons. the number of maximum electrons that can occupy one shell is defined by the formula of 2nî or 2n square, where n is the number of the shell.
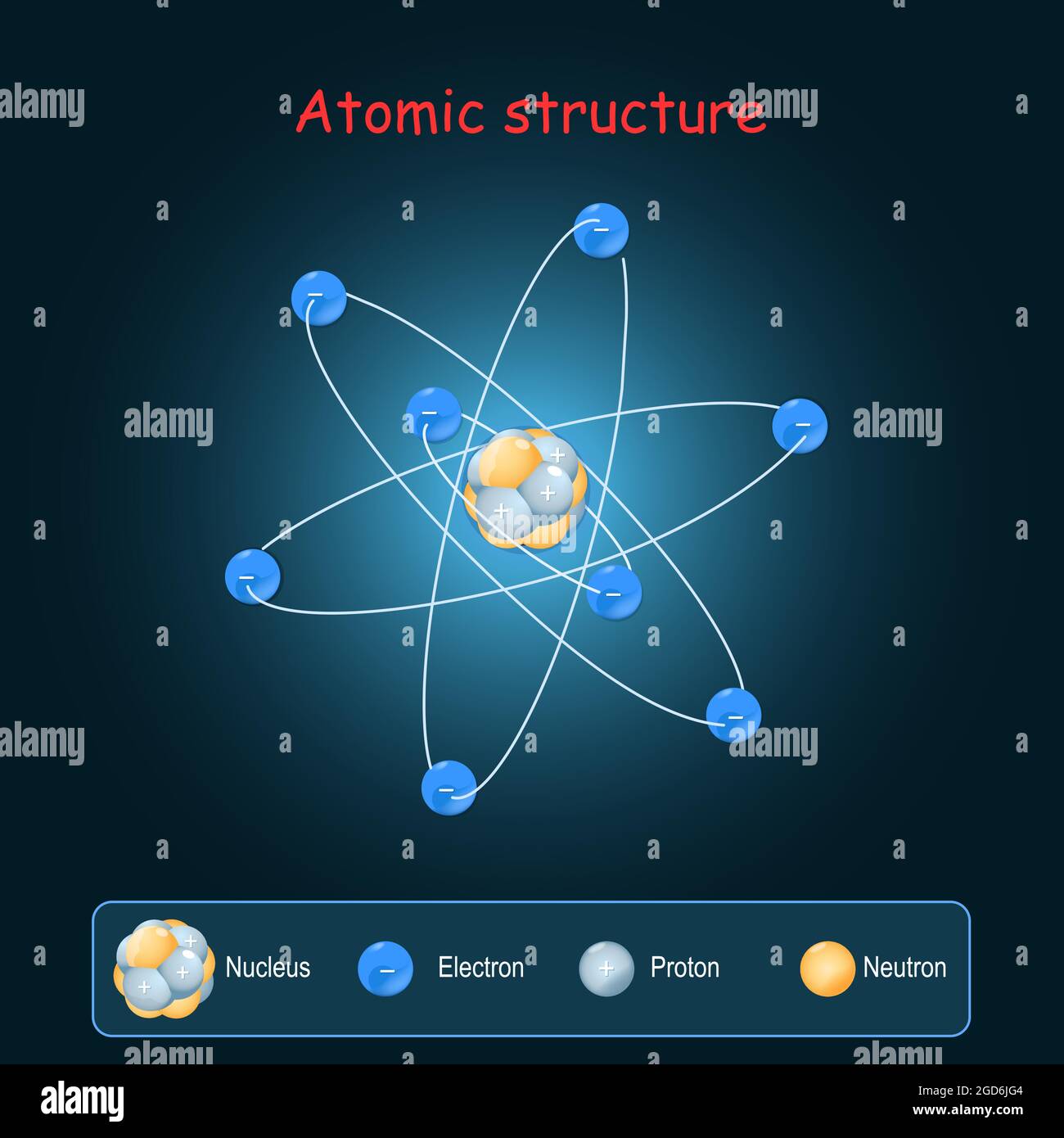
Atomic Structure Electrons And Nucleus With Neutrons And Protons Chemical elements are made up of only one type of atom—gold contains only gold atoms, and neon contains only neon atoms and they are ranked in order of their atomic number (the total number of protons in its nucleus) in a chart called the periodic table. If one is oriented along the x axis, called 2 p x, the second is along the y axis, called 2 p y, and the third is along the z axis, called 2 p z, as illustrated in figure 1.2.3. The development of modern atomic theory revealed much about the inner structure of atoms. it was learned that an atom contains a very small nucleus composed of positively charged protons and uncharged neutrons, surrounded by a much larger volume of space containing negatively charged electrons. These levels may be designated k l m n shells (or 1 2 3 4 orbits). the first level contains only 2 electrons. the second level can hold 6 electrons. the number of maximum electrons that can occupy one shell is defined by the formula of 2nî or 2n square, where n is the number of the shell.
Comments are closed.