What Is A Clinical Study Mcrc
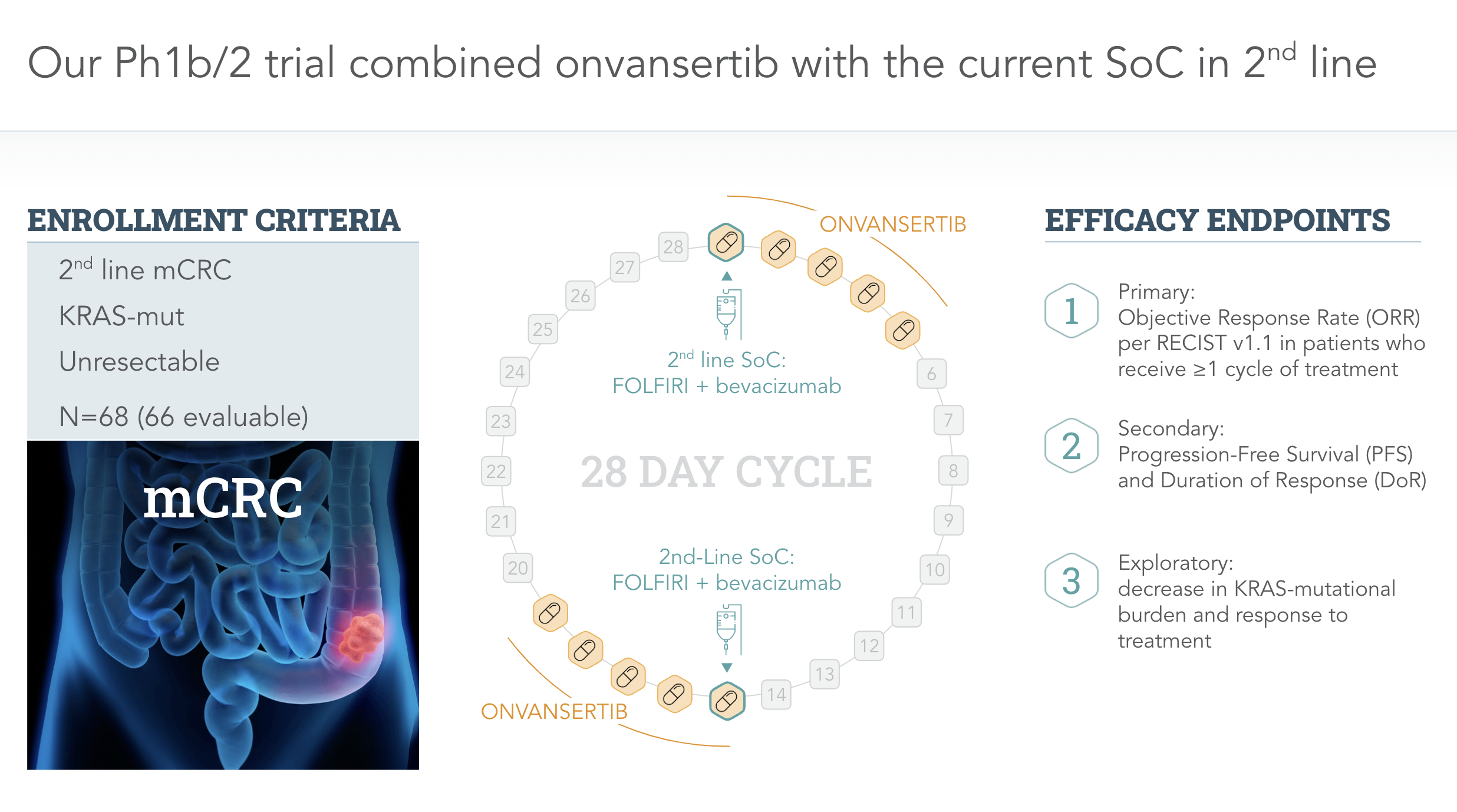
Mcrc Clinical Program Cardiff Oncology Currently, there is no consensus definition for clinically meaningful outcomes in randomized clinical trials (rcts) designed to evaluate new treatments for patients with refractory metastatic colorectal cancer (mcrc). This is an observational, retrospective, international and multicenter study funded by fondazione oncologia niguarda ets to assess the efficacy of anti egfr rechallenge regimens in the largest real world cohort of mss mcrc patients screened for ras braf pik3ca ctdna status on liquid biopsy within the men1611 02 c precise 01 trial. the study will collect molecular, clinical and outcome data of.
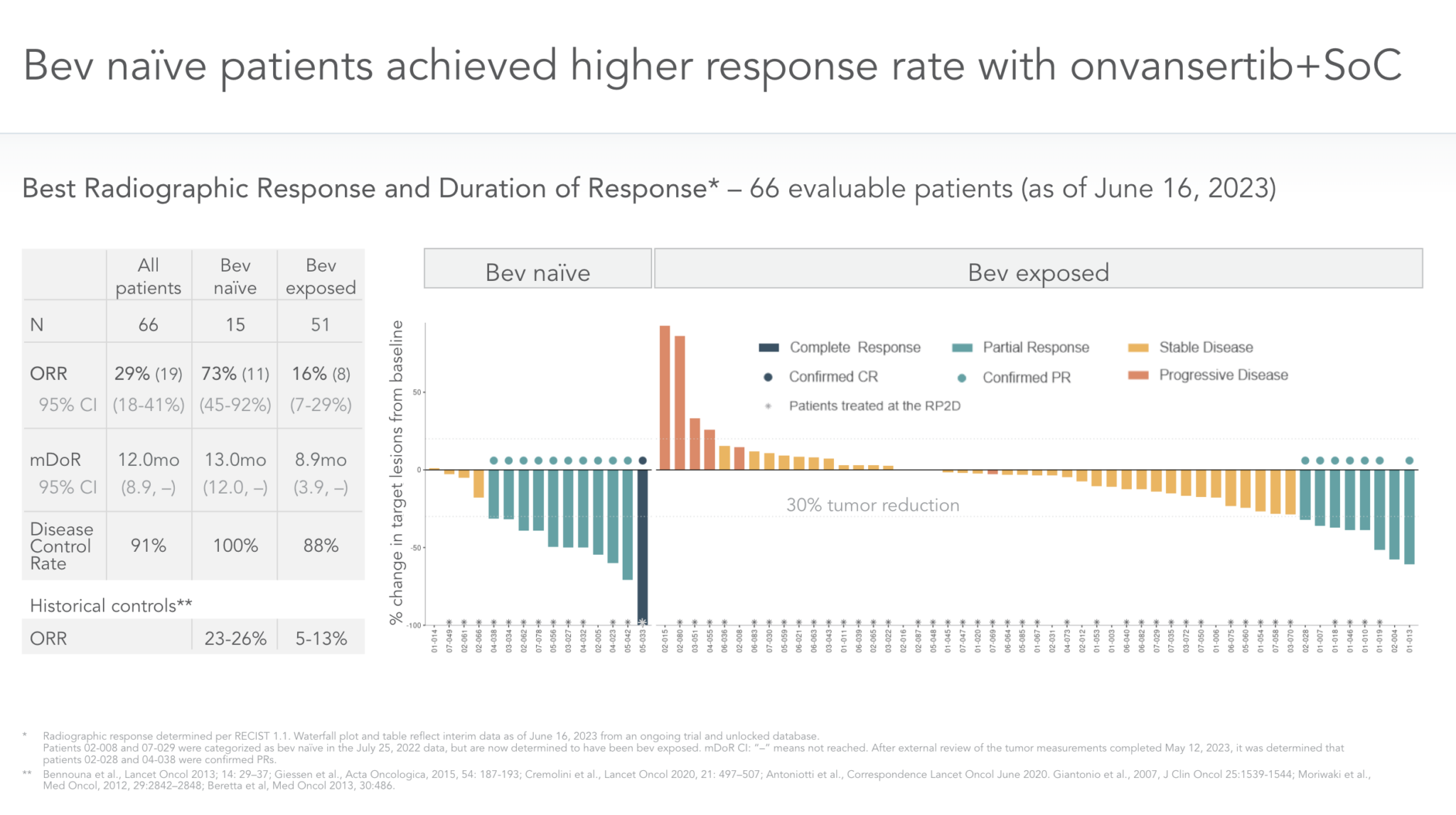
Mcrc Clinical Program Cardiff Oncology Precemtabart tocentecan (m9140), the first anti ceacam5 adc with an exatecan payload (topoisomerase 1 inhibitor), showed a predictable, manageable safety profile and promising early clinical activity in the dose escalation of the phase 1 proceade crc 01 study (nct05464030) in heavily pretreated patients with mcrc. Clinical studies, such as the trial conducted by hurwitz et al., evaluated the efficacy of bevacizumab when added to standard therapy. in this trial, which included irinotecan, fluorouracil, and leucovorin (ifl), the addition of bevacizumab resulted in a significant improvement in pfs. Abstract. background: arfolitixorin ([6r] 5,10 methylene tetrahydrofolate) is a direct acting folate in development that may enhance treatment outcomes in patients with mcrc by bypassing the multi step metabolic activation required by leucovorin (lv), the most commonly used folate agent. in a phase 3 study with bevacizumab (nct03750786), arfox (arfolitixorin, 5fu, oxaliplatin) demonstrated. This is an observational, retrospective, international and multicenter study funded by fondazione oncologia niguarda ets to assess the efficacy of anti egfr rechallenge regimens in the largest real world cohort of mss mcrc patients screened for ras braf pik3ca ctdna status on liquid biopsy within the men1611 02 c precise 01 trial. the study will collect molecular, clinical and outcome data of.
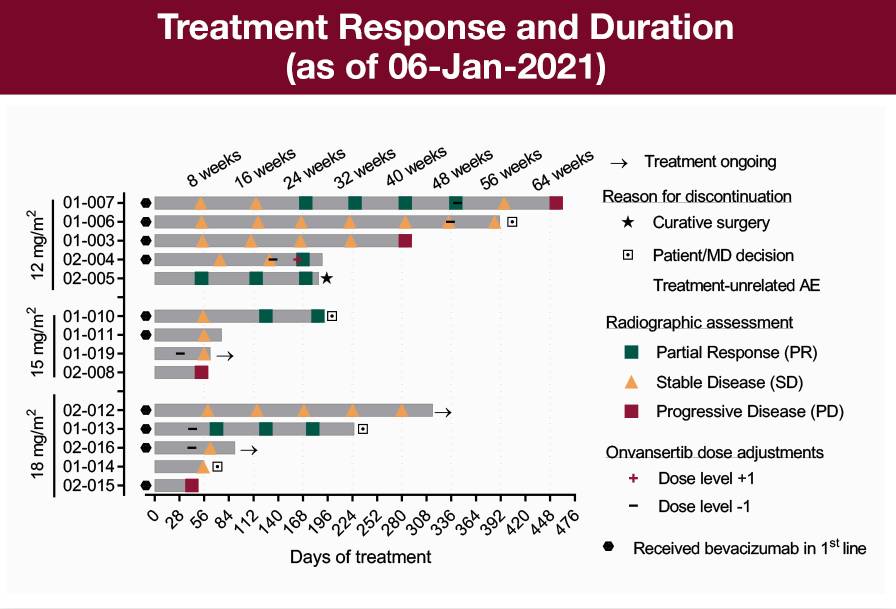
Kras Mutated Mcrc Clinical Program Cardiff Oncology Abstract. background: arfolitixorin ([6r] 5,10 methylene tetrahydrofolate) is a direct acting folate in development that may enhance treatment outcomes in patients with mcrc by bypassing the multi step metabolic activation required by leucovorin (lv), the most commonly used folate agent. in a phase 3 study with bevacizumab (nct03750786), arfox (arfolitixorin, 5fu, oxaliplatin) demonstrated. This is an observational, retrospective, international and multicenter study funded by fondazione oncologia niguarda ets to assess the efficacy of anti egfr rechallenge regimens in the largest real world cohort of mss mcrc patients screened for ras braf pik3ca ctdna status on liquid biopsy within the men1611 02 c precise 01 trial. the study will collect molecular, clinical and outcome data of. The open label, single center, non randomized phase 2 trial utilizes a simon two stage design and includes three disease cohorts: metastatic colorectal cancer, cholangiocarcinoma, and adrenocortical cancer. the publication reports data from the metastatic colorectal cancer cohort of the trial. key findings from stage 1 (n=9) of the 22 patient study* in colorectal cancer patients with liver. Mcrc has been conducting clinical trials since 1984 with a primary focus in rheumatology. our research site includes a 12 bed phase i unit with a highly trained staff of phase i. In the case of clinical or radiological suspicion of mcrc, histological diagnosis of colorectal adenocarcinoma or related metastases should always be confirmed before administration of anticancer therapy [iv, a].
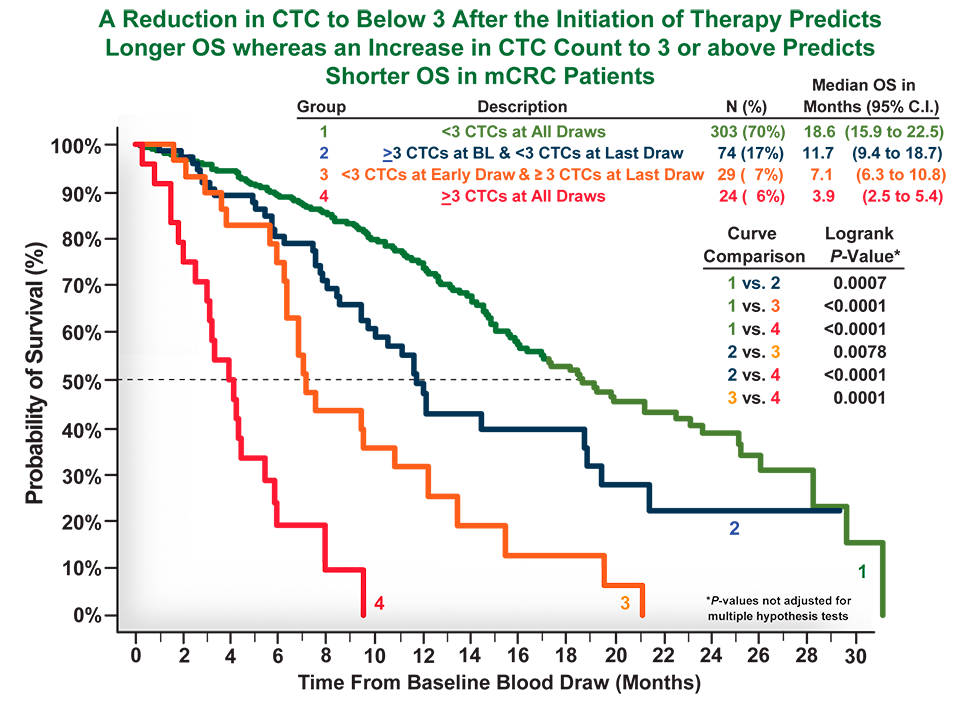
Cellsearch Mcrc Clinical Trials Case Studies The open label, single center, non randomized phase 2 trial utilizes a simon two stage design and includes three disease cohorts: metastatic colorectal cancer, cholangiocarcinoma, and adrenocortical cancer. the publication reports data from the metastatic colorectal cancer cohort of the trial. key findings from stage 1 (n=9) of the 22 patient study* in colorectal cancer patients with liver. Mcrc has been conducting clinical trials since 1984 with a primary focus in rheumatology. our research site includes a 12 bed phase i unit with a highly trained staff of phase i. In the case of clinical or radiological suspicion of mcrc, histological diagnosis of colorectal adenocarcinoma or related metastases should always be confirmed before administration of anticancer therapy [iv, a].
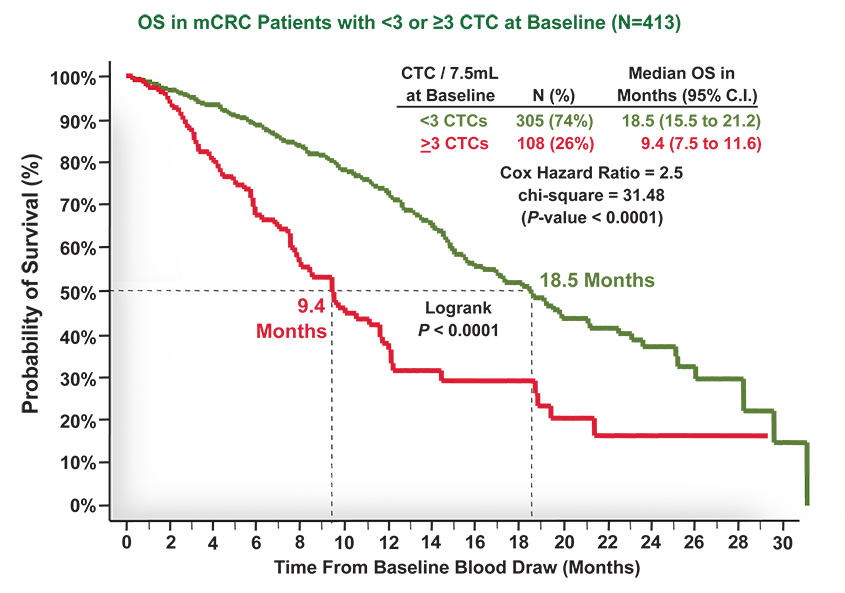
Cellsearch Mcrc Clinical Trials Case Studies In the case of clinical or radiological suspicion of mcrc, histological diagnosis of colorectal adenocarcinoma or related metastases should always be confirmed before administration of anticancer therapy [iv, a].
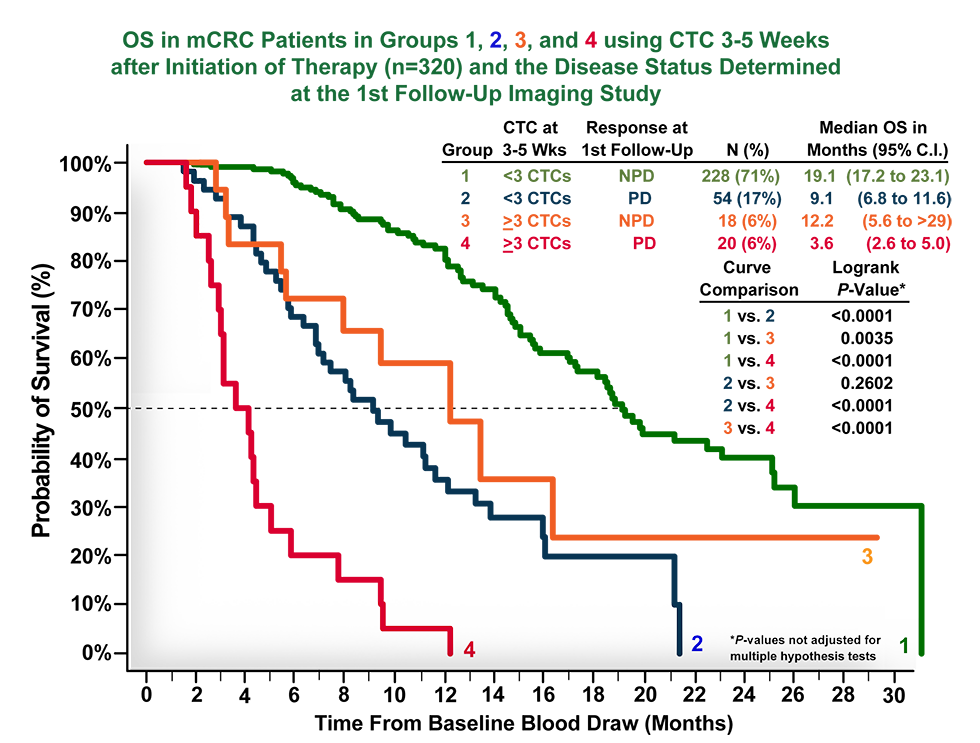
Cellsearch Mcrc Clinical Trials Case Studies
Comments are closed.