What Do Atoms Look Like

Eli5 How Do We Know How Atoms Look Like R Explainlikeimfive However, atoms are made up of three types of subatomic particles: the heavy protons and neutrons that make up the nucleus (the central part of the atom), and the much lighter electrons that circle the nucleus in orbital paths called shells. Real pictures of atoms. discover how stm, afm, sem & tem let us see atoms for the first time. learn how these images revolutionized science.

The Internet Is Obsessed With This Cute Way To Visualize Atoms Atoms are extremely small, typically around 100 picometers across. a human hair is about a million carbon atoms wide. atoms are smaller than the shortest wavelength of visible light, which means humans cannot see atoms with conventional microscopes. Learn how different microscopy techniques can reveal the structure and properties of atoms and molecules. see examples of electron microscopy, atomic force microscopy, scanning tunneling microscopy, and more. There are 94 natural elements and 24 artificial elements that exist today. an atom’s size is tiny, with a diameter of 0.1 to 0.5 nanometers (1 × 10 −10 to 5 × 10 −10 m). thus, they cannot be seen with our naked eye. a layer of an atom is somewhat similar to a sheet of paper. Atoms cannot be seen with the naked eye, or even under a standard microscope. an atom is too small to deflect visible light waves, meaning it will not show up under light focusing microscopes.

The Simple Atomic Model Ks3 Chemistry Revision There are 94 natural elements and 24 artificial elements that exist today. an atom’s size is tiny, with a diameter of 0.1 to 0.5 nanometers (1 × 10 −10 to 5 × 10 −10 m). thus, they cannot be seen with our naked eye. a layer of an atom is somewhat similar to a sheet of paper. Atoms cannot be seen with the naked eye, or even under a standard microscope. an atom is too small to deflect visible light waves, meaning it will not show up under light focusing microscopes. Most atoms have three different subatomic particles inside them: protons, neutrons, and electrons. The appearance of the atom is often simplified for teaching purposes using the classical bohr model. this model depicts the nucleus at the center, with electrons orbiting it in fixed, circular paths or shells, much like planets orbiting the sun. The atomic number and mass number do not directly tell you the number of electrons in an atom. however, atoms have the same number of electrons as protons, so the atomic number also gives the. Learn about the history, models, and structure of atoms, the smallest particles of matter. find out what atoms look like under a microscope and how they are composed of subatomic particles.
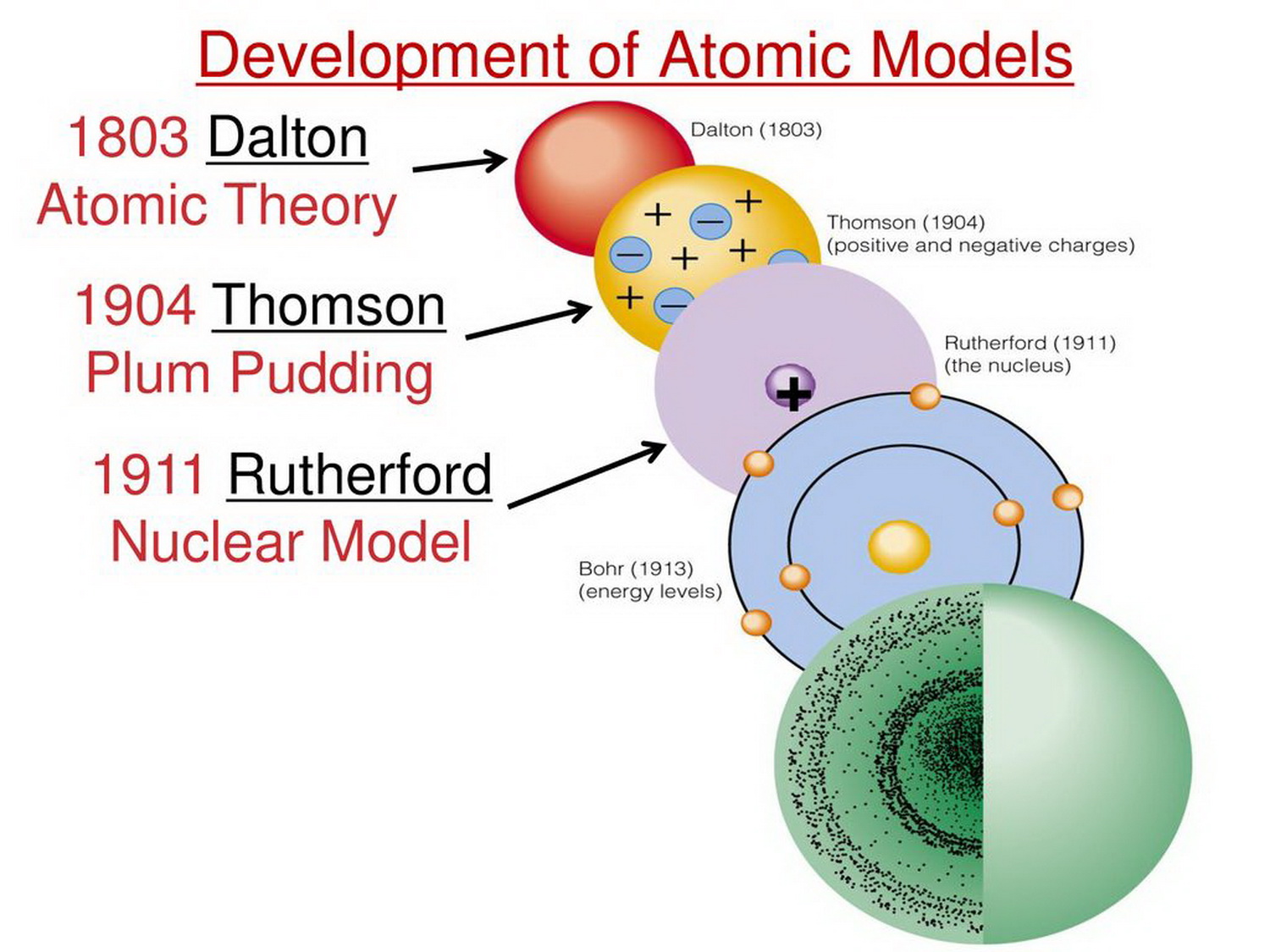
Ib Chemistry Sl Hl 2 1 Atomic Model Most atoms have three different subatomic particles inside them: protons, neutrons, and electrons. The appearance of the atom is often simplified for teaching purposes using the classical bohr model. this model depicts the nucleus at the center, with electrons orbiting it in fixed, circular paths or shells, much like planets orbiting the sun. The atomic number and mass number do not directly tell you the number of electrons in an atom. however, atoms have the same number of electrons as protons, so the atomic number also gives the. Learn about the history, models, and structure of atoms, the smallest particles of matter. find out what atoms look like under a microscope and how they are composed of subatomic particles.
Comments are closed.