What Are Clinical Trial Phases

3 Phases Of Clinical Trial Pdf Researchers design clinical trials to answer specific research questions related to a medical product. these trials follow a specific study plan, called a protocol, that is developed by the. Clinical trials happen in four phases. researchers use each phase to determine if the treatment is safe and effective. if your healthcare team recommends that you participate in a clinical trial,.
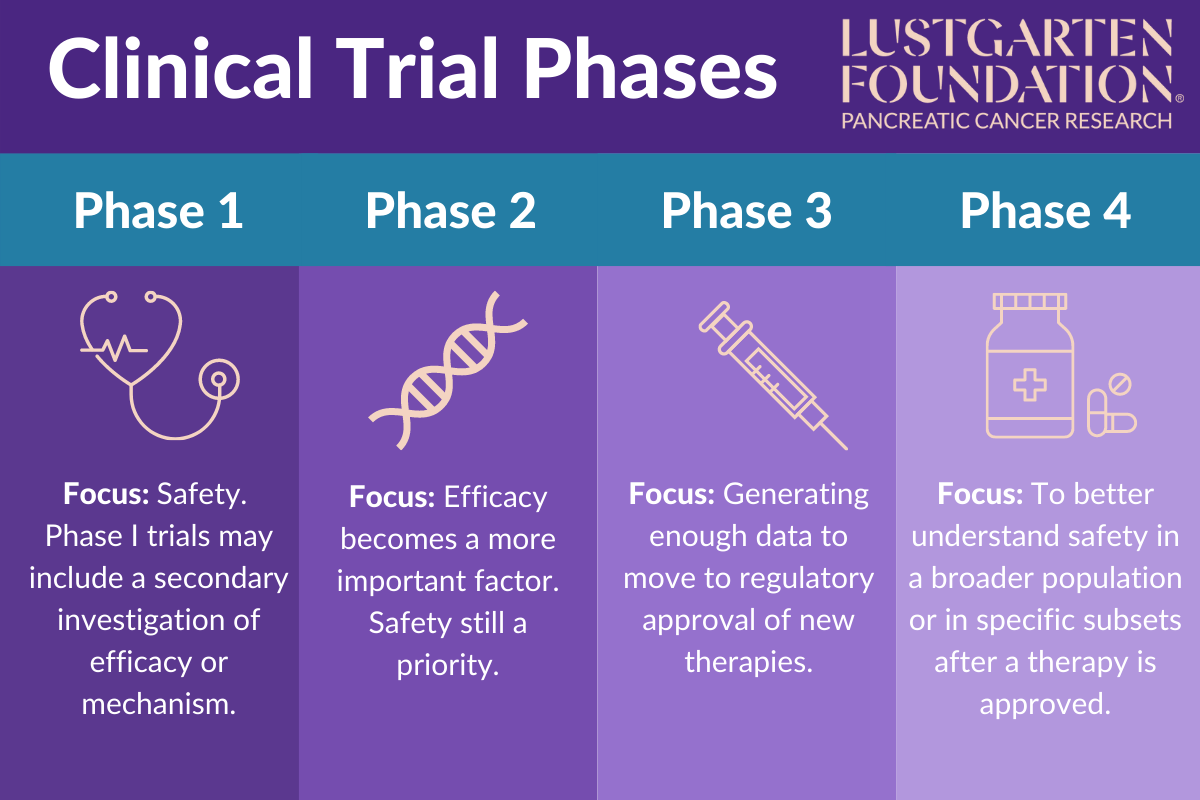
Clinical Trial Phases Clinical trials are the scientific cornerstone of modern medicine, providing the evidence base for the safety and efficacy of new drugs and therapies. they are traditionally organized into four successive phases – i, ii, iii, and iv – each with distinct objectives, designs, and regulatory roles. Phase 0 and phase 2 are called exploratory trial phases, phase 1 is termed the non therapeutic phase, phase 3 is known as the therapeutic confirmatory phase, and phase 4 is called the post approval or the post marketing surveillance phase. Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays. The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives.
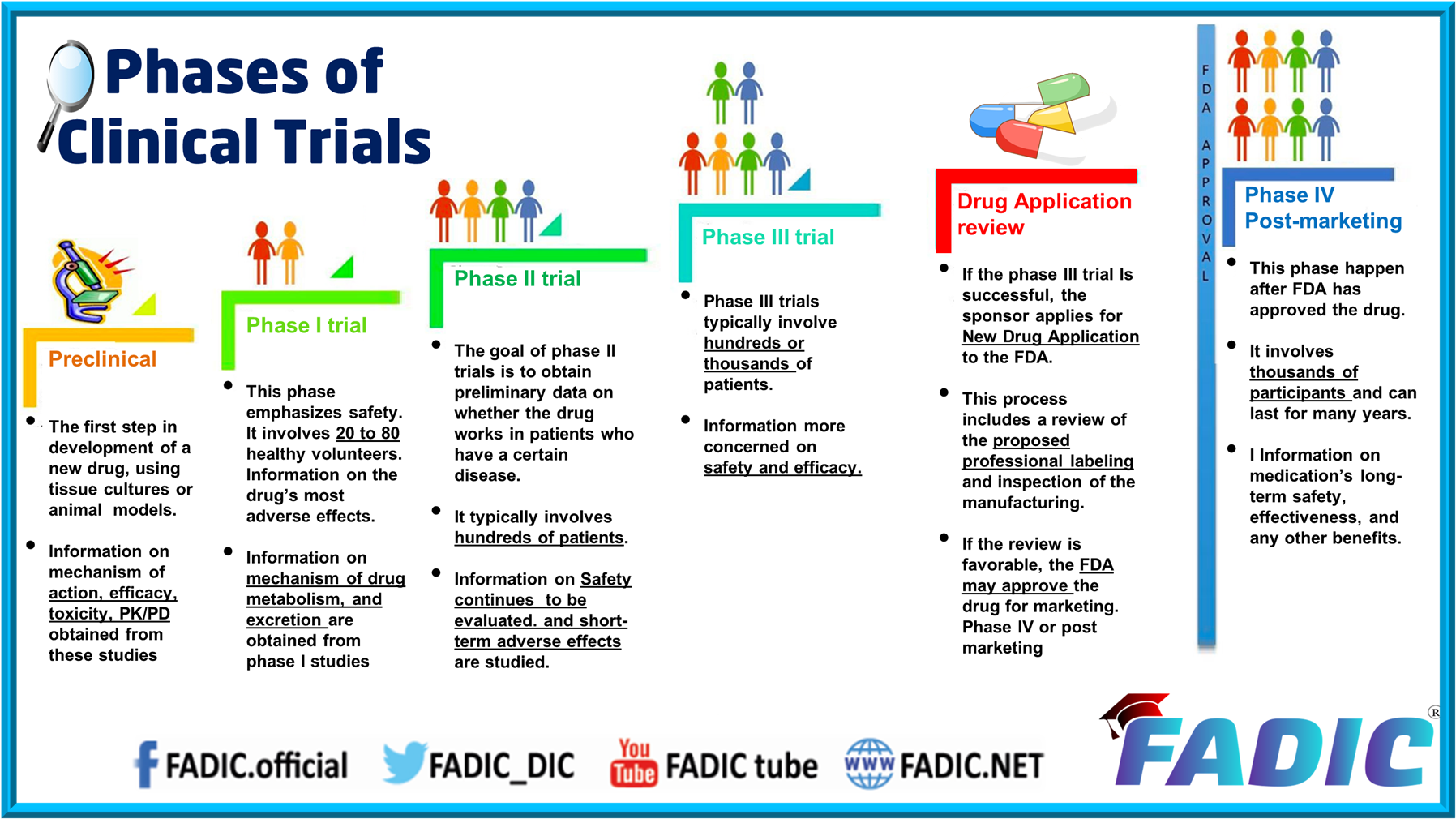
Fadic Calender Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays. The 4 phases of clinical trials, from initial safety testing to post market monitoring. learn how each stage progresses, participant numbers, and objectives. There are 4 clinical trials phases, each with its specific goal. phase 1 ensures safety, while phase 2 studies effectiveness. phase 3 confirms large scale success, and phase 4 monitors real world safety. every phase minimizes risks before new treatments reach patients. Clinical trials are required before any new medicine can be approved for public use. this guide explains all clinical trial phases, from laboratory research to phase 4 monitoring, in simple terms. learn how safety, effectiveness, and approval are tested step by step. Clinical trials are divided into different phases. most trials are phase 1, 2 or 3. but some trials are classified as phase 0 and some as phase 4. What is a clinical trial phase? the journey of a new medical intervention is meticulously mapped through various clinical trial phases. these stages are designed to answer critical questions related to a drug’s pharmacology, therapeutic effect, dosage requirements, and long term safety.

Clinical Trial Phases What Happens In Phase 1 2 3 And 4 Velocity There are 4 clinical trials phases, each with its specific goal. phase 1 ensures safety, while phase 2 studies effectiveness. phase 3 confirms large scale success, and phase 4 monitors real world safety. every phase minimizes risks before new treatments reach patients. Clinical trials are required before any new medicine can be approved for public use. this guide explains all clinical trial phases, from laboratory research to phase 4 monitoring, in simple terms. learn how safety, effectiveness, and approval are tested step by step. Clinical trials are divided into different phases. most trials are phase 1, 2 or 3. but some trials are classified as phase 0 and some as phase 4. What is a clinical trial phase? the journey of a new medical intervention is meticulously mapped through various clinical trial phases. these stages are designed to answer critical questions related to a drug’s pharmacology, therapeutic effect, dosage requirements, and long term safety.

Understand Different Clinical Trial Phases Types Clinical trials are divided into different phases. most trials are phase 1, 2 or 3. but some trials are classified as phase 0 and some as phase 4. What is a clinical trial phase? the journey of a new medical intervention is meticulously mapped through various clinical trial phases. these stages are designed to answer critical questions related to a drug’s pharmacology, therapeutic effect, dosage requirements, and long term safety.
Comments are closed.