Water Molecules Part1
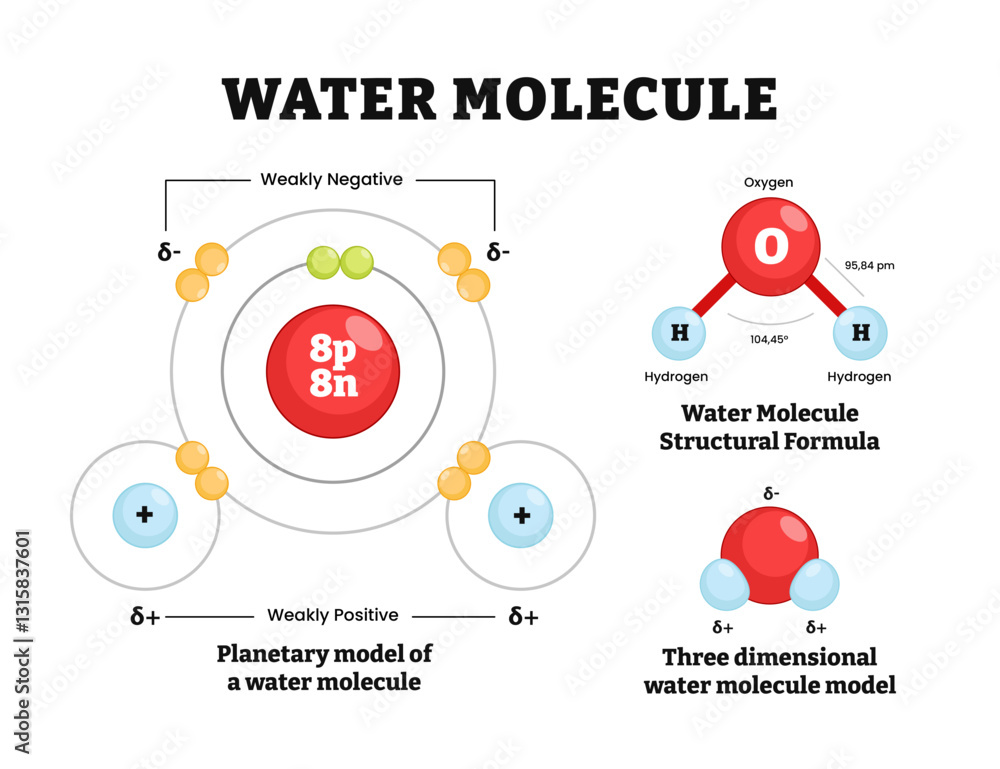
Water Molecule Structure Diagram Vector Illustration Hydrogen And Project name: e content for undergraduate students of science in graduate programmes project investigator: dr. mandira sikdar module name: water molecules part 1 more. Water chemistry part1 free download as pdf file (.pdf), text file (.txt) or view presentation slides online. the document discusses the essential properties of water, including its polar nature, hydrogen bonding, and unique behaviors such as cohesion, adhesion, and high specific heat.

Ppt Unit 4 The Hydrosphere Powerpoint Presentation Free Download Water is formed when one oxygen atom forms a single covalent bond with two separate hydrogen atoms. this arrangement gives oxygen the two electrons it needs to fill its outer shell and allows both hydrogen atoms to receive the single electrons they need for their outer shells. Water (h₂o) – definition, structure, preparation, uses, benefits water stands as a paramount covalent compound within the realm of chemistry. this molecule is composed of two hydrogen atoms bonded to a single oxygen atom through covalent bonds, a configuration that renders it essential for myriad biological processes and ecological systems. Each water molecule is made up of one oxygen and two hydrogen atoms. overall, each water molecule has a partial negative charge near the oxygen and partial positive charge at each hydrogen – figure 1. This page explores the molecular characteristics and importance of water, highlighting its composition of one oxygen and two hydrogen atoms, its bent shape due to polar covalent bonding, and its ….
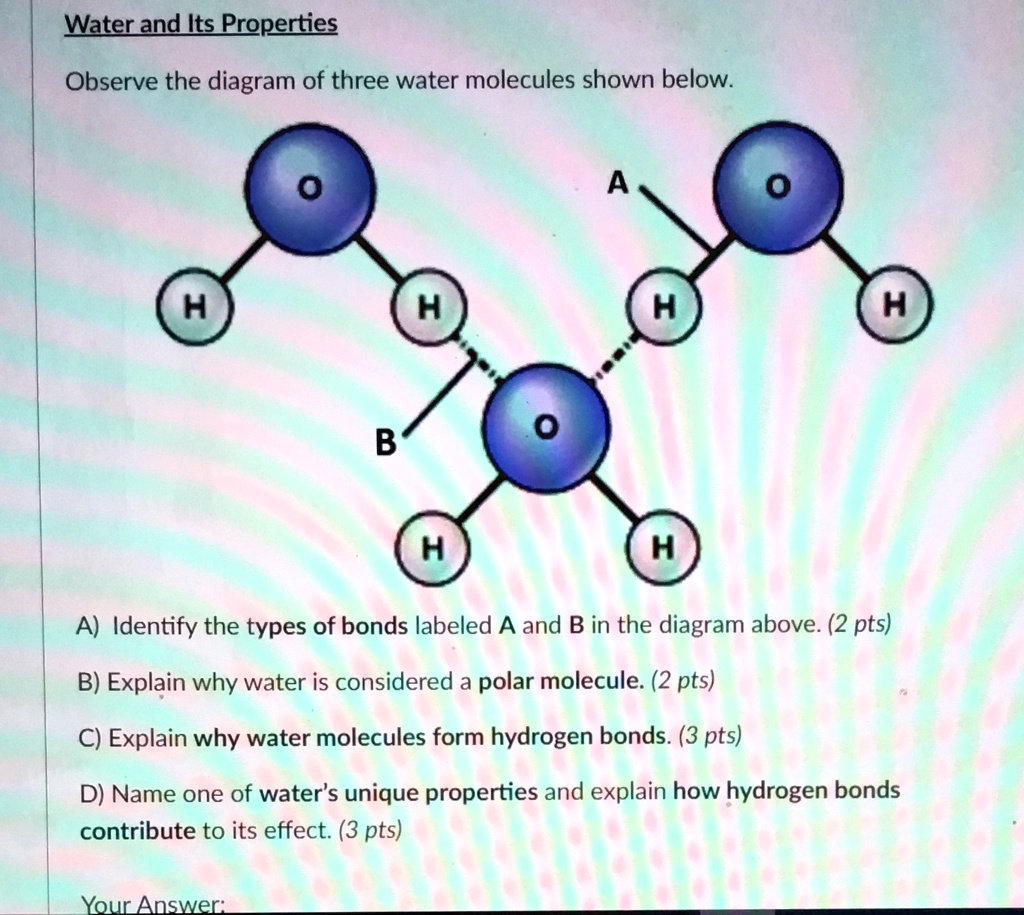
Solved Water And Its Properties Observe The Diagram Of Three Water Each water molecule is made up of one oxygen and two hydrogen atoms. overall, each water molecule has a partial negative charge near the oxygen and partial positive charge at each hydrogen – figure 1. This page explores the molecular characteristics and importance of water, highlighting its composition of one oxygen and two hydrogen atoms, its bent shape due to polar covalent bonding, and its …. Polarity of water molecules the key to understanding water’s chemical behavior is its molecular structure. a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. this is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. Water is a molecular compound consisting of polar molecules that have a bent shape. the oxygen atom acquires a partial negative charge while the hydrogen atom acquires a partial positive charge. Recall: water (h 2 o) is a compound consisting of only two elements: one oxygen atom and two hydrogen atoms. oxygen is more electronegative than hydrogen; therefore, polar covalent bonds hold the atoms within water molecules together. The chemical formula for water is h 2 o which indicates that a single molecule of water is made up of two hydrogen atoms and one oxygen atom. the atoms in a water molecule are connected to each other by polar covalent bonds.

Science Online The Importance Of The Water And Its Structure Polarity of water molecules the key to understanding water’s chemical behavior is its molecular structure. a water molecule consists of two hydrogen atoms bonded to an oxygen atom, and its overall structure is bent. this is because the oxygen atom, in addition to forming bonds with the hydrogen atoms, also carries two pairs of unshared electrons. Water is a molecular compound consisting of polar molecules that have a bent shape. the oxygen atom acquires a partial negative charge while the hydrogen atom acquires a partial positive charge. Recall: water (h 2 o) is a compound consisting of only two elements: one oxygen atom and two hydrogen atoms. oxygen is more electronegative than hydrogen; therefore, polar covalent bonds hold the atoms within water molecules together. The chemical formula for water is h 2 o which indicates that a single molecule of water is made up of two hydrogen atoms and one oxygen atom. the atoms in a water molecule are connected to each other by polar covalent bonds.
Comments are closed.