Water Molecule Structure
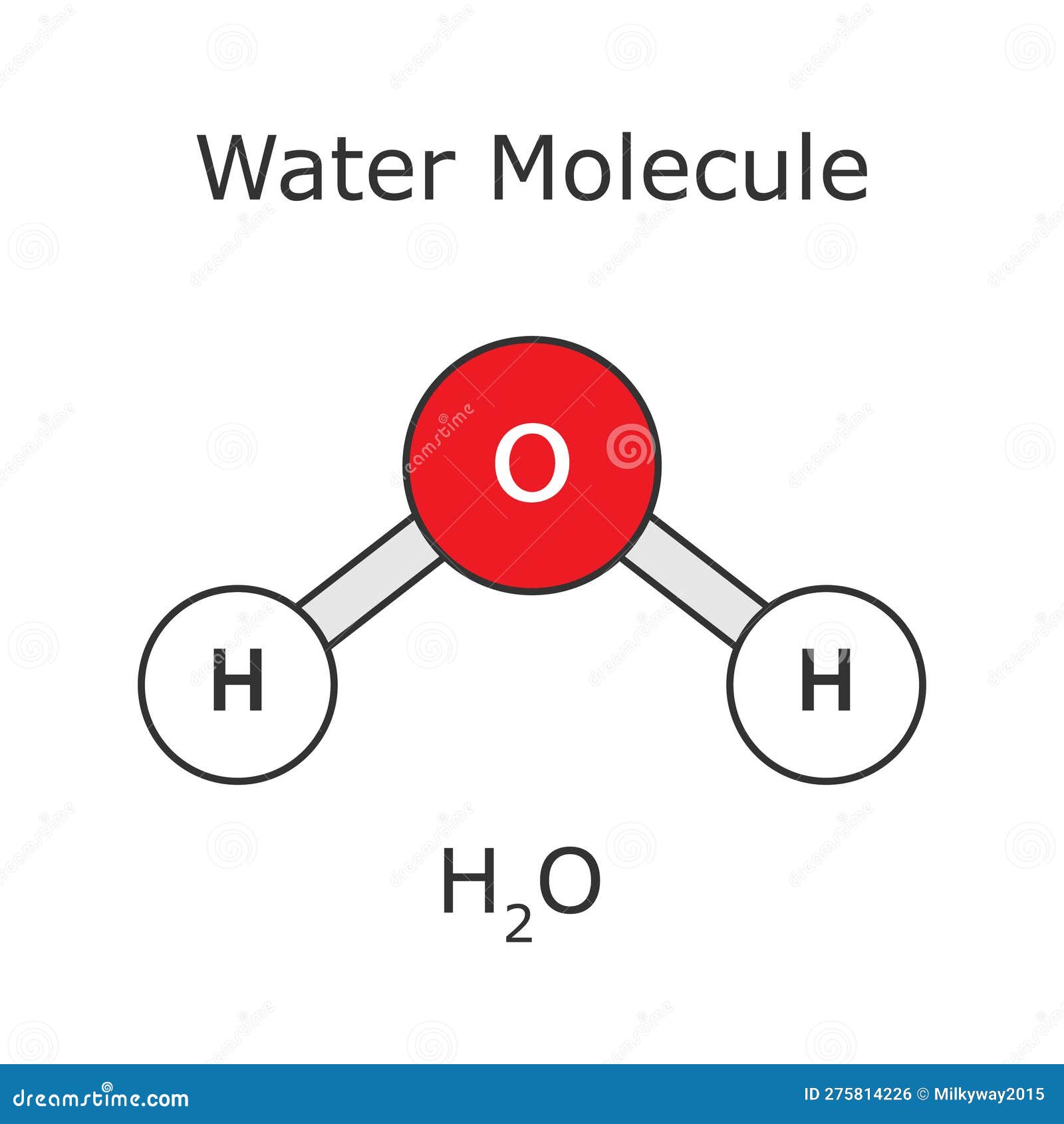
Water Molecule Molecule Structure Atomic H2o Stock Vector Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). As a chemical compound, a water molecule contains one oxygen and two hydrogen atoms that are connected by covalent bonds. water is a liquid at standard ambient temperature and pressure, but it often co exists on earth with its solid state, ice; and gaseous state, steam (water vapor).
/illustration-of-properties-and-molecular-structure-of-water-consisting-of-two-hydrogen-atoms-covalently-bonded-to-single-oxygen-atom-85594579-583b267c5f9b58d5b1b9e84b.jpg)
Water Molecule Structure Water, being a polar molecule, undergoes strong intermolecular hydrogen bonding which is a large contributor to its physical and chemical properties. [20] it is vital for all known forms of life, despite not providing food energy or being an organic micronutrient. This review, aiming at a broader general audience, attempts to review briefly some research trends related to water's structure, properties and applications. new experimental results for the debated phenomena of water bridge and exclusion zone are also presented and discussed. The structure of liquid water is believed to consist of aggregates of water molecules that form and re form continually. this short range order, as it is called, accounts for other unusual properties of water, such as its high viscosity and surface tension. Water is a tiny bent molecule with the molecular formula h 2 o, consisting of two light hydrogen atoms attached to each 16 fold heavier oxygen atom. each molecule is electrically neutral but polar, with the center of positive and negative charges located in different places.

Biв Olв Oв Gy Bд л г Lй Jд Structure Of A Water Molecule The structure of liquid water is believed to consist of aggregates of water molecules that form and re form continually. this short range order, as it is called, accounts for other unusual properties of water, such as its high viscosity and surface tension. Water is a tiny bent molecule with the molecular formula h 2 o, consisting of two light hydrogen atoms attached to each 16 fold heavier oxygen atom. each molecule is electrically neutral but polar, with the center of positive and negative charges located in different places. Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). Learn about the molecular structure, shape, charge distribution, electronic structure and van der waals radius of water. see diagrams, parameters and calculations based on ab initio and experimental data. The molecular structure of water molecule remains a cornerstone concept in chemistry, biology, and environmental science. its distinctive bent geometry, polar nature, and capacity for hydrogen bonding collectively explain the exceptional properties of water that pervade natural and engineered systems. as analytical techniques evolve, so too does our understanding of this vital molecule. Learn how to draw the lewis structure of water (h2o) easily. complete step by step explanation, formal charges, molecular geometry, and bond angles.
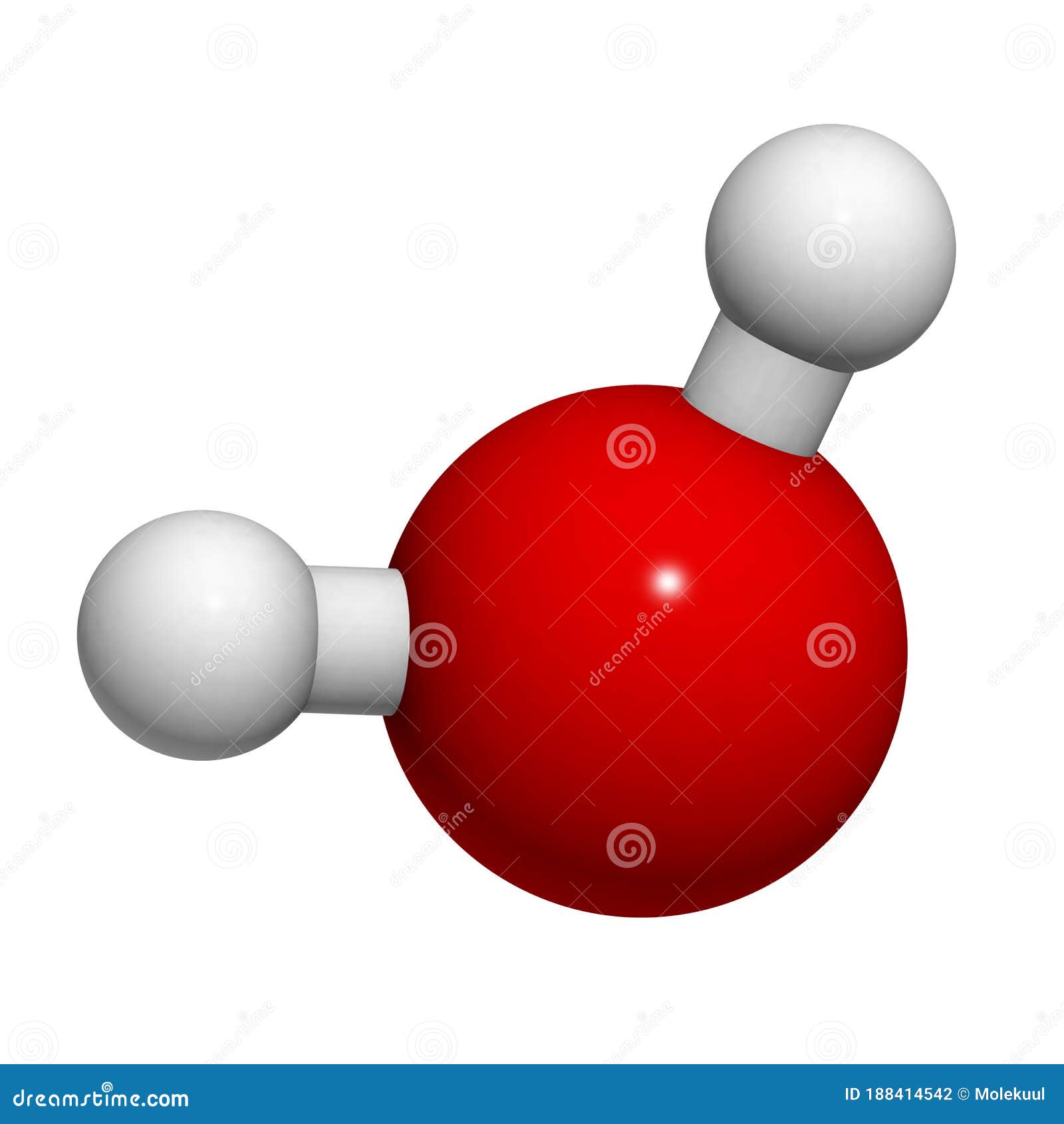
Water Molecule Chemical Structure Royalty Free Stock Image Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. because of the higher electronegativity of the oxygen atom, the bonds are polar covalent (polar bonds). Learn about the molecular structure, shape, charge distribution, electronic structure and van der waals radius of water. see diagrams, parameters and calculations based on ab initio and experimental data. The molecular structure of water molecule remains a cornerstone concept in chemistry, biology, and environmental science. its distinctive bent geometry, polar nature, and capacity for hydrogen bonding collectively explain the exceptional properties of water that pervade natural and engineered systems. as analytical techniques evolve, so too does our understanding of this vital molecule. Learn how to draw the lewis structure of water (h2o) easily. complete step by step explanation, formal charges, molecular geometry, and bond angles.
Comments are closed.