Validation And Verification Design Verification Validation For

Design Verification Pdf Verification And Validation Formal Verification is used to make sure that the design has addressed every requirement, while validation is used to prove that the design can meet the requirements set out for it. While design verification and design validation serve different purposes, they are equally vital to the product development process. verification ensures that the product aligns with technical specifications, while validation confirms that the product meets end user needs and is fit for its intended purpose.
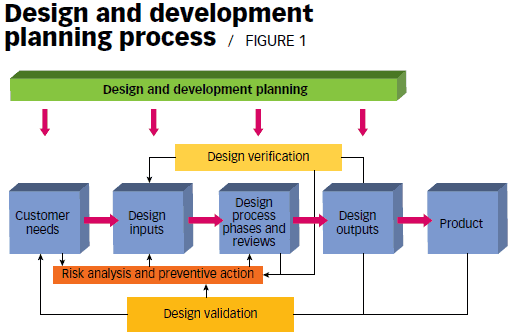
Design Validation Verification Services The Next Rex Learn exactly what design verification and design validation are, how they are the same, how they are different, and best practices for medical devices. This article will further explain the key differences between design verification and design validation, and will guide you on how to implement these in your operational processes. Before diving into technical details, it is important to understand the fundamental purpose of design verification and validation in any regulated product development setting. design verification is a formal process to ensure that the design outputs are consistent with the design inputs. In the analysis of medical device performance and regulatory adherence, the terms “verification” and “validation” (often abbreviated as v&v) are frequently conflated.

Design Verification Vs Design Validation Emma International Before diving into technical details, it is important to understand the fundamental purpose of design verification and validation in any regulated product development setting. design verification is a formal process to ensure that the design outputs are consistent with the design inputs. In the analysis of medical device performance and regulatory adherence, the terms “verification” and “validation” (often abbreviated as v&v) are frequently conflated. Verification ensures products meet specifications and design requirements, while validation confirms they satisfy user needs and operational requirements under real world conditions. The objective evidence needed for a validation is the result of a test or other form of determination such as performing alternative calculations or reviewing documents. the word “validated” is used to designate the corresponding status. the use conditions for validation can be real or simulated. What is design verification and validation? design verification ensures that a design meets all specified requirements set out at the beginning of a development process. it answers the question, "did we build the product right?". Design verification ensures a device meets design specifications, while validation confirms it meets user needs and intended use. verification focuses on technical accuracy; validation ensures real world performance and regulatory compliance.
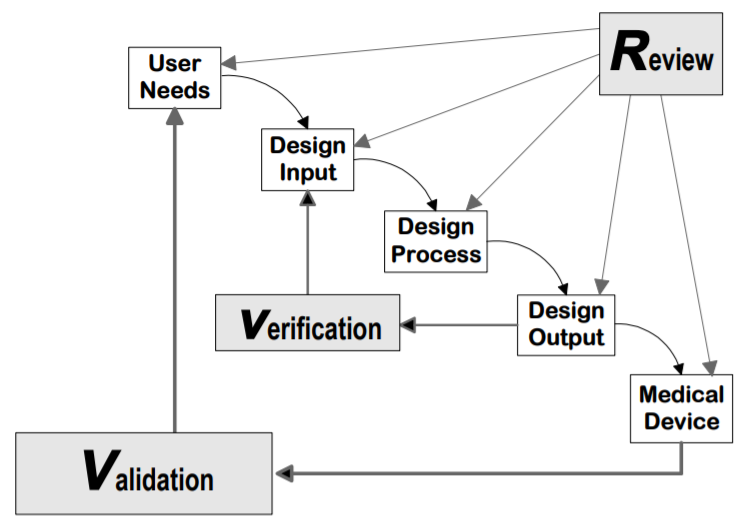
Analysis Of Design Validation And Verification For Medical Device Human Verification ensures products meet specifications and design requirements, while validation confirms they satisfy user needs and operational requirements under real world conditions. The objective evidence needed for a validation is the result of a test or other form of determination such as performing alternative calculations or reviewing documents. the word “validated” is used to designate the corresponding status. the use conditions for validation can be real or simulated. What is design verification and validation? design verification ensures that a design meets all specified requirements set out at the beginning of a development process. it answers the question, "did we build the product right?". Design verification ensures a device meets design specifications, while validation confirms it meets user needs and intended use. verification focuses on technical accuracy; validation ensures real world performance and regulatory compliance.

Design Verification And Validation Procedure What is design verification and validation? design verification ensures that a design meets all specified requirements set out at the beginning of a development process. it answers the question, "did we build the product right?". Design verification ensures a device meets design specifications, while validation confirms it meets user needs and intended use. verification focuses on technical accuracy; validation ensures real world performance and regulatory compliance.

Design Verification Vs Design Validation Fda Requirements
Comments are closed.