Understanding Radioactive Decay Physics

Nuclear Stability And Radioactive Decay Physics Radioactive decay happens at a predictable rate for large groups of atoms, and half life is the measure that captures this behavior. it tells us how long it takes for half of a sample to decay, allowing us to model and calculate changes in radioactive materials over time. Radioactive decay occurs for all nuclei with z> 82, and also for some unstable isotopes with z <83. the decay rate is proportional to the number of original (undecayed) nuclei n in a substance.

Radioactive Decay Basics Types Effects When an individual nucleus transforms into another with the emission of radiation, the nucleus is said to decay. radioactive decay occurs for all nuclei. Explore the fundamentals of radioactive decay, including alpha, beta, and gamma emissions, half life concepts, and practical applications in science and industry. The radioactive decay law, uses the properties of radioactive substances to estimate the age of a substance. radioactive carbon has the same chemistry as stable carbon, so it mixes into the ecosphere and eventually becomes part of every living organism. Having defined the basic parameters responsible for nuclear stability, let us explore the physics of radioactive decay which governs what happens to unstable nuclei.
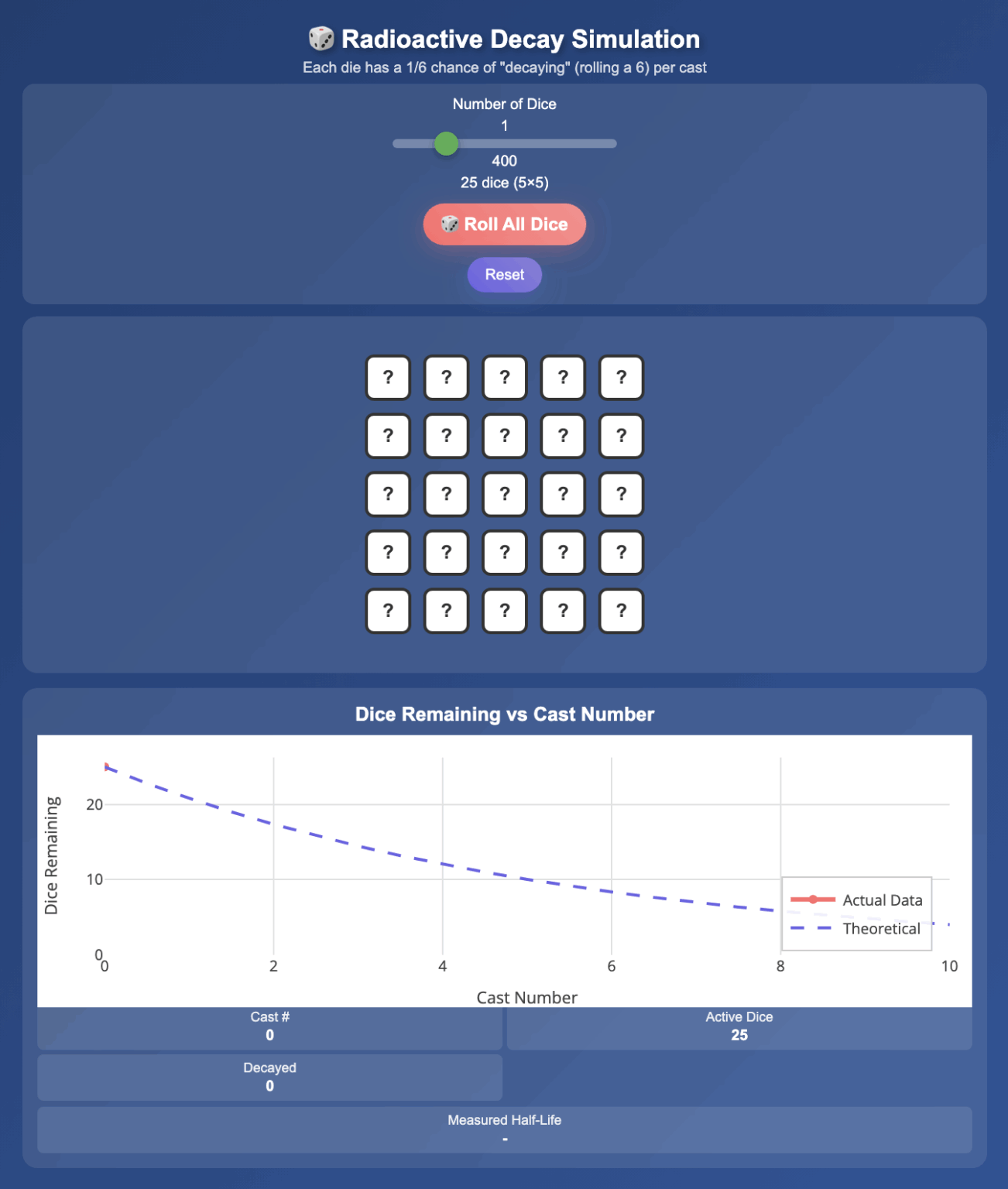
Simulation Radioactive Decay Physics Lens The radioactive decay law, uses the properties of radioactive substances to estimate the age of a substance. radioactive carbon has the same chemistry as stable carbon, so it mixes into the ecosphere and eventually becomes part of every living organism. Having defined the basic parameters responsible for nuclear stability, let us explore the physics of radioactive decay which governs what happens to unstable nuclei. The nuclei of some isotopes are not everlasting. they spit out bits and pieces of themselves in decay processes — alpha, beta, gamma, neutron, and others. Radioactive decay is a fundamental nuclear process that helps us understand how unstable atomic nuclei transform to achieve greater stability. this article explores the various types of radioactive decay and their unique characteristics. Radioactive decay is a fundamental process by which an unstable atomic nucleus loses energy by emitting radiation. this natural phenomenon is a cornerstone in fields such as nuclear physics, geology, and even medicine. So in short, what is radioactivity or what is radioactive decay? it's a process in which you have an unstable nucleus that spontaneously changes to a new, more stable nucleus and, in doing so, it releases high energy, ionizing radiation.
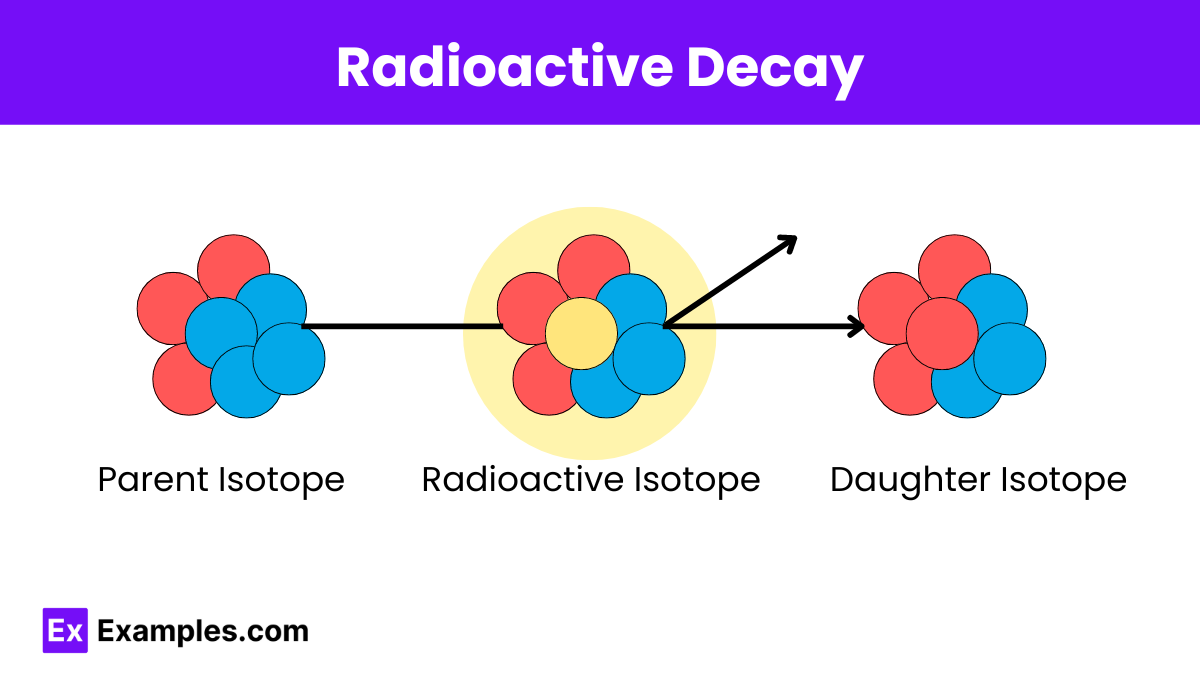
Radioactive Decay Law Examples Derivation Uses Faq S The nuclei of some isotopes are not everlasting. they spit out bits and pieces of themselves in decay processes — alpha, beta, gamma, neutron, and others. Radioactive decay is a fundamental nuclear process that helps us understand how unstable atomic nuclei transform to achieve greater stability. this article explores the various types of radioactive decay and their unique characteristics. Radioactive decay is a fundamental process by which an unstable atomic nucleus loses energy by emitting radiation. this natural phenomenon is a cornerstone in fields such as nuclear physics, geology, and even medicine. So in short, what is radioactivity or what is radioactive decay? it's a process in which you have an unstable nucleus that spontaneously changes to a new, more stable nucleus and, in doing so, it releases high energy, ionizing radiation.

Unit 7 2 Radioactive Decay Notes Practice Questions Ap Physics Radioactive decay is a fundamental process by which an unstable atomic nucleus loses energy by emitting radiation. this natural phenomenon is a cornerstone in fields such as nuclear physics, geology, and even medicine. So in short, what is radioactivity or what is radioactive decay? it's a process in which you have an unstable nucleus that spontaneously changes to a new, more stable nucleus and, in doing so, it releases high energy, ionizing radiation.
Comments are closed.