Understanding Periodic Table Groups Properties Elements And Course

Periodic Table Groups Names And Properties Discover the 18 periodic table groups of elements, their names, examples, key properties, and how groups differ from families and periods. This interactive periodic table of element groups arranges the chemical elements according to periodicity or common properties.
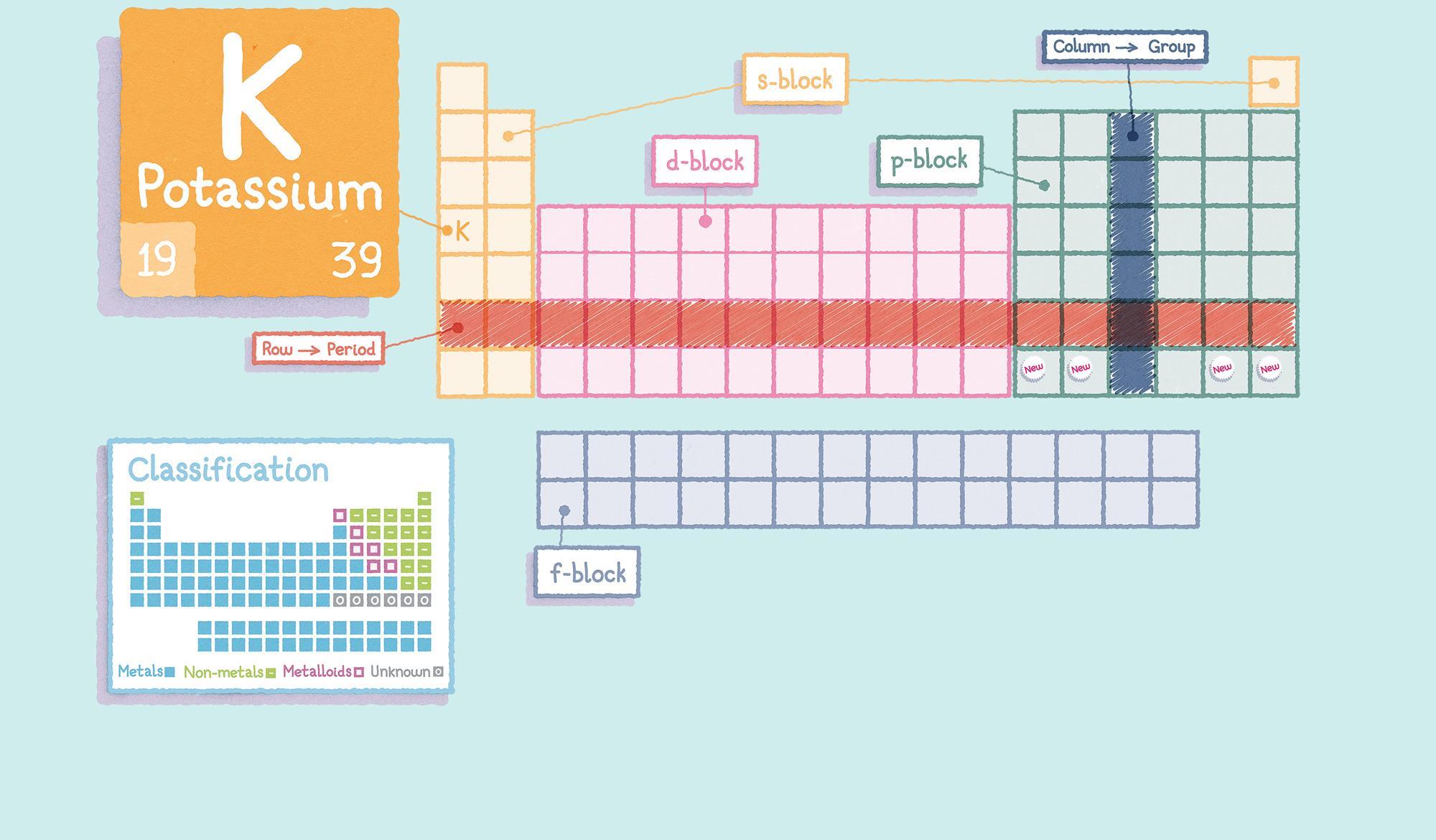
Periodic Table Of Elements Groups The properties of elements in the same group of the periodic system are, although similar, not identical. the trend in properties from the lighter to the heavier elements may be attributed to changes in the strength of binding of the outer electrons and especially to the increasing size of the atoms. Discover how groups of elements are organized and understand the significance of each group, from alkali metals to noble gases, including trends in physical and chemical properties, applications, and environmental implications. Learn how chemistry’s periodic table reveals patterns of reactivity and properties. understand groups, periods, metals, nonmetals, and metalloids. Periods, groups, and families of the periodic table defined with a labeled diagram. how many are there. what are their names. learn their properties with examples.
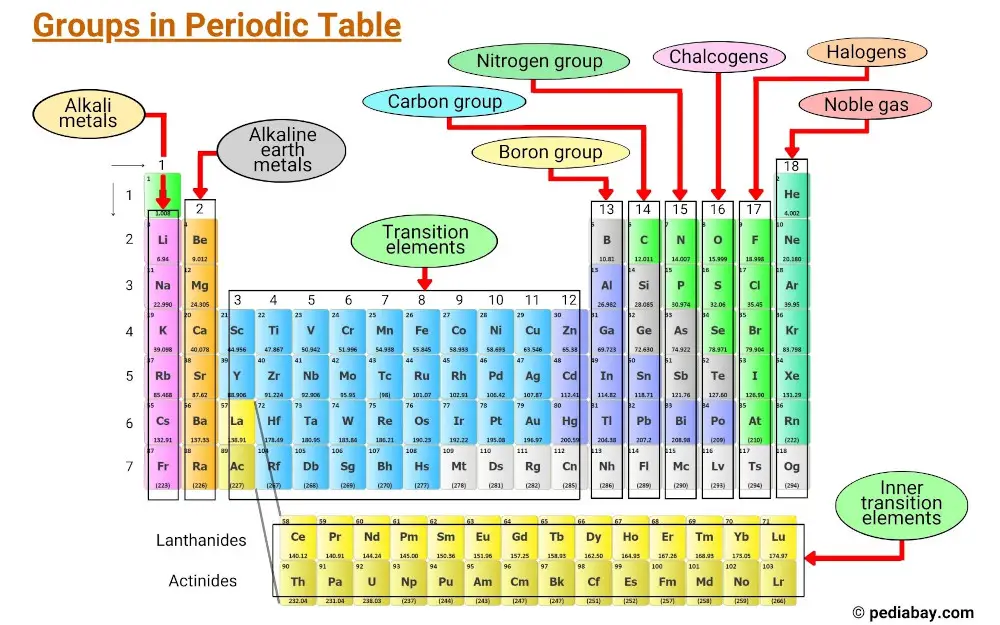
Periodic Table Of Elements Groups Learn how chemistry’s periodic table reveals patterns of reactivity and properties. understand groups, periods, metals, nonmetals, and metalloids. Periods, groups, and families of the periodic table defined with a labeled diagram. how many are there. what are their names. learn their properties with examples. Although most modern periodic tables are arranged in eighteen groups (columns) of elements, mendeleev's original periodic table had the elements organized into eight groups and twelve periods (rows). on the periodic table, elements that have similar properties are in the same groups (vertical). Learn what the periodic table is, how elements are grouped, periodic trends, and why it matters. download charts and tips for chemistry exam success. Explore the modern periodic table. learn about its structure, including the seven periods and eighteen groups, and understand the properties of elements. Classification of elements into groups and development of periodic law and periodic table are the consequences of systematising the knowledge gained by a number of scientists through their observations and experiments. the german chemist, johann dobereiner in early 1800’s was the first to consider the idea of trends among properties of elements. by 1829 he noted a similarity among the.

Periodic Table Of Elements Groups Although most modern periodic tables are arranged in eighteen groups (columns) of elements, mendeleev's original periodic table had the elements organized into eight groups and twelve periods (rows). on the periodic table, elements that have similar properties are in the same groups (vertical). Learn what the periodic table is, how elements are grouped, periodic trends, and why it matters. download charts and tips for chemistry exam success. Explore the modern periodic table. learn about its structure, including the seven periods and eighteen groups, and understand the properties of elements. Classification of elements into groups and development of periodic law and periodic table are the consequences of systematising the knowledge gained by a number of scientists through their observations and experiments. the german chemist, johann dobereiner in early 1800’s was the first to consider the idea of trends among properties of elements. by 1829 he noted a similarity among the.
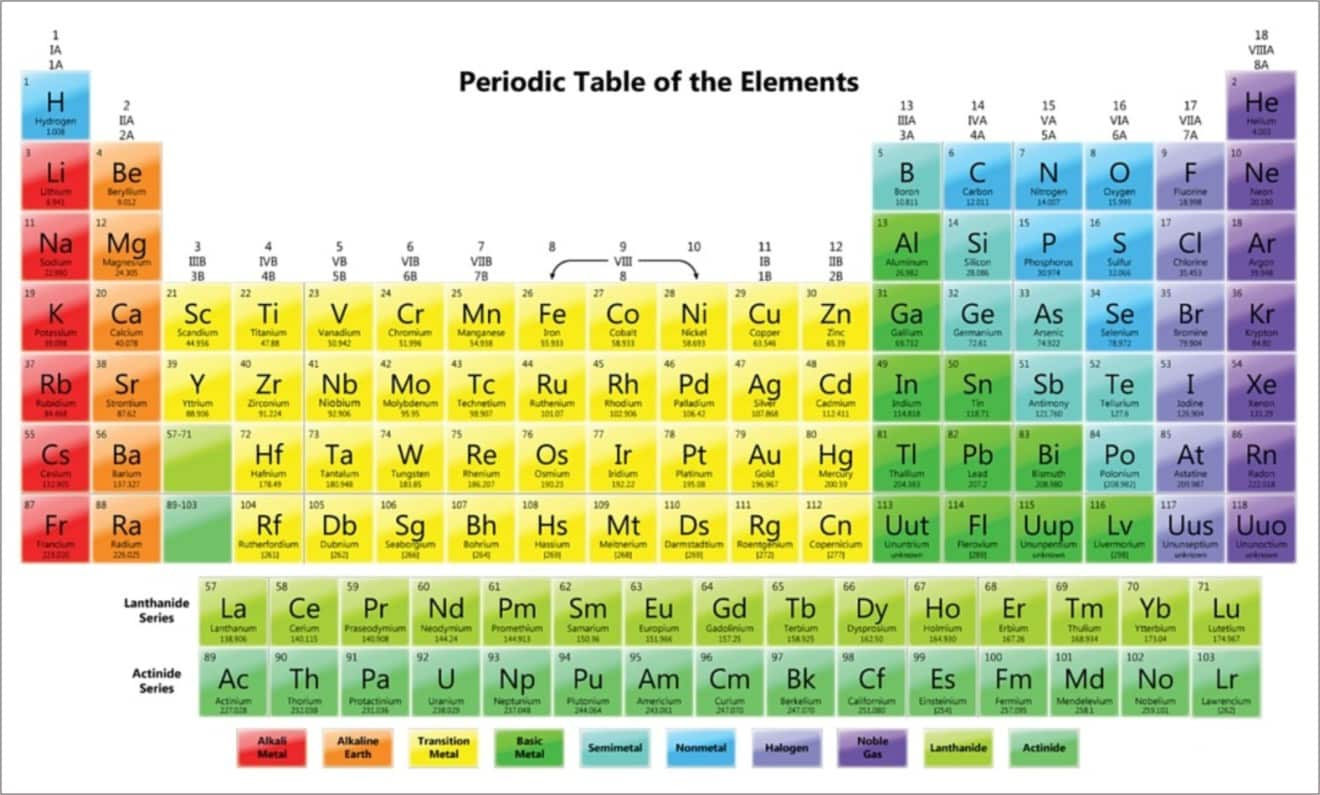
Periodic Table Groups Definition Cabinets Matttroy Explore the modern periodic table. learn about its structure, including the seven periods and eighteen groups, and understand the properties of elements. Classification of elements into groups and development of periodic law and periodic table are the consequences of systematising the knowledge gained by a number of scientists through their observations and experiments. the german chemist, johann dobereiner in early 1800’s was the first to consider the idea of trends among properties of elements. by 1829 he noted a similarity among the.
Comments are closed.