Understanding Ions And Ionic Charge

Ionic Compounds Consist Of Charge Ions Join us as we demystify ions and ionic charge step by step, unraveling the mysteries of charged particles in chemical reactions. we'll begin by introducing the concept of ions and their. Chapter 3: ions, ionic compounds, and nomenclature. an ion is a small particle having an electrical charge. ions are either single, charged atoms (simple ions), or small charged “molecules” (polyatomic ions). examples of simple ions are na , ca 2, cl ‐, and s ‐2. examples of polyatomic ions are nh.
Understanding Ions And Ionic Bonds In Chemistry Video Summary By An ion is an atom or a molecule that carries a net electrical charge because its total number of electrons is unequal to its total number of protons. while a neutral atom maintains a perfect balance between its positive and negative components, an ion represents a state of electrical imbalance. Atoms or groups of atoms that possess a net electrical charge are called ions; they can have either a positive charge (cations) or a negative charge (anions). ions can consist of one atom (monatomic ions) or several (polyatomic ions). Electrons can move from one atom to another; when they do, species with overall electric charges are formed. such species are called ions. species with overall positive charges are termed cations, while species with overall negative charges are called anions. Ions are atoms that have a positive or negative charge because they have unequal numbers of protons and electrons. if atoms lose electrons, they become positive ions, or cations. if atoms gain electrons, they become negative ions, or anions. consider the example of fluorine (see figure below).

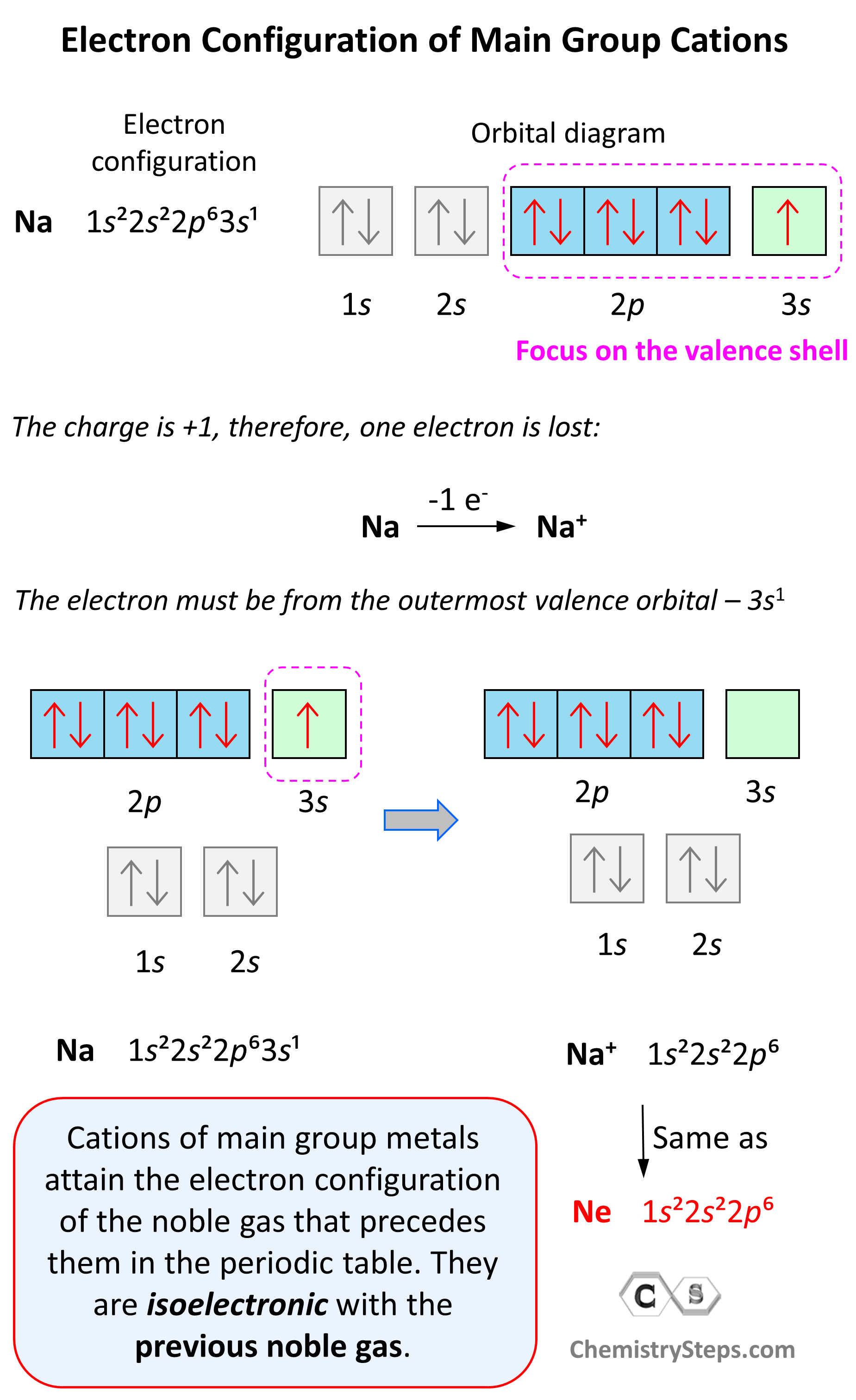
Hydrogen Ionic Charge Electrons can move from one atom to another; when they do, species with overall electric charges are formed. such species are called ions. species with overall positive charges are termed cations, while species with overall negative charges are called anions. Ions are atoms that have a positive or negative charge because they have unequal numbers of protons and electrons. if atoms lose electrons, they become positive ions, or cations. if atoms gain electrons, they become negative ions, or anions. consider the example of fluorine (see figure below). Demystify the charge on the ion! learn how ionic charges are determined, their importance in chemistry, and real world applications. get clear answers now!. Ionic charge refers to the electrical charge possessed by an ion, which is an atom or molecule that has gained or lost one or more electrons. the ionic charge determines the chemical properties and reactivity of the ion. Ions form when electrons are transferred between atoms, creating charged particles. because these particles carry electric charge, they interact through attraction and repulsion. Recall that atoms carrying positive or negative charges are called ions. if an atom has gained one or more electrons, it is negatively charged and is called an anion. if an atom has lost one or more electrons, it is positively charged and is called a cation.
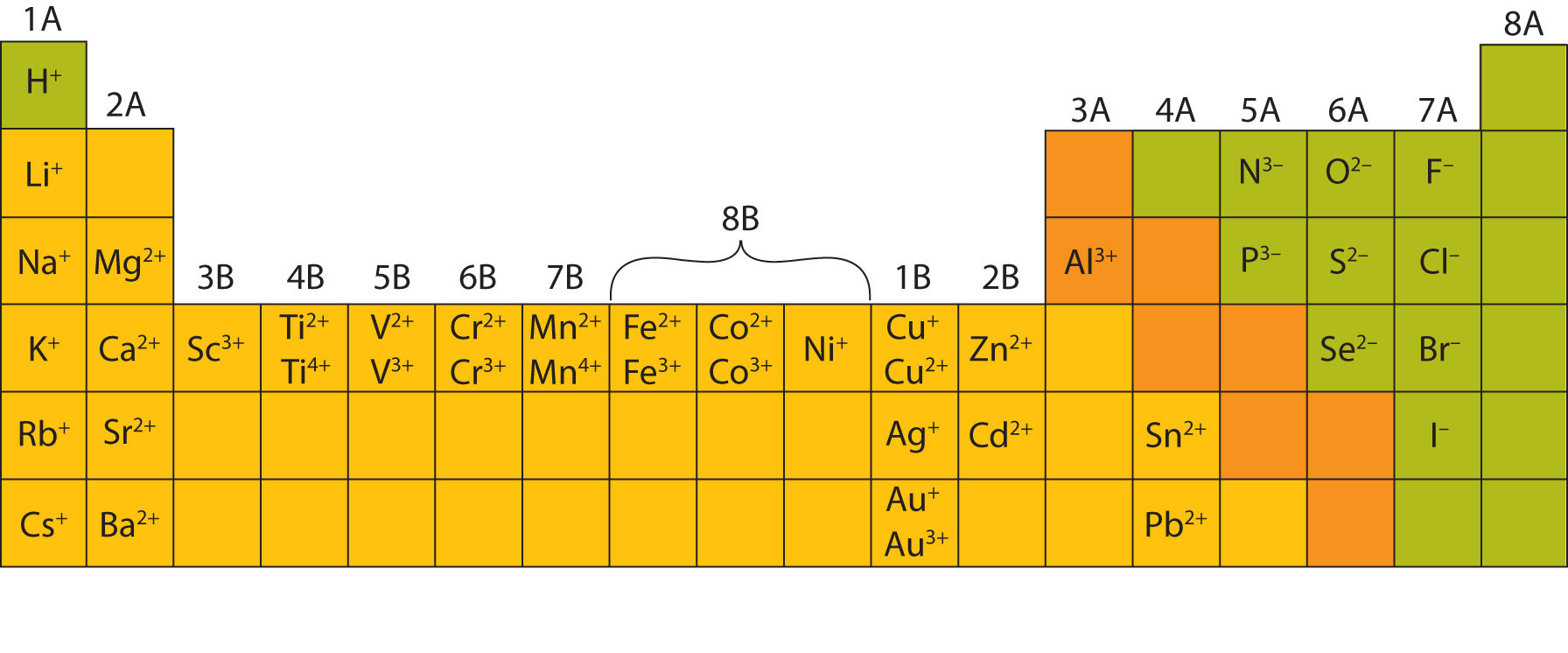
Ionic Charge Periodic Table Ukrainerd Demystify the charge on the ion! learn how ionic charges are determined, their importance in chemistry, and real world applications. get clear answers now!. Ionic charge refers to the electrical charge possessed by an ion, which is an atom or molecule that has gained or lost one or more electrons. the ionic charge determines the chemical properties and reactivity of the ion. Ions form when electrons are transferred between atoms, creating charged particles. because these particles carry electric charge, they interact through attraction and repulsion. Recall that atoms carrying positive or negative charges are called ions. if an atom has gained one or more electrons, it is negatively charged and is called an anion. if an atom has lost one or more electrons, it is positively charged and is called a cation.

Understanding Ions And Ionic Compounds Chem 101 Lecture Notes Ions form when electrons are transferred between atoms, creating charged particles. because these particles carry electric charge, they interact through attraction and repulsion. Recall that atoms carrying positive or negative charges are called ions. if an atom has gained one or more electrons, it is negatively charged and is called an anion. if an atom has lost one or more electrons, it is positively charged and is called a cation.
Ionic Charge Pattern Periodic Table Infoupdate Org
Comments are closed.