Understanding Informed Consent
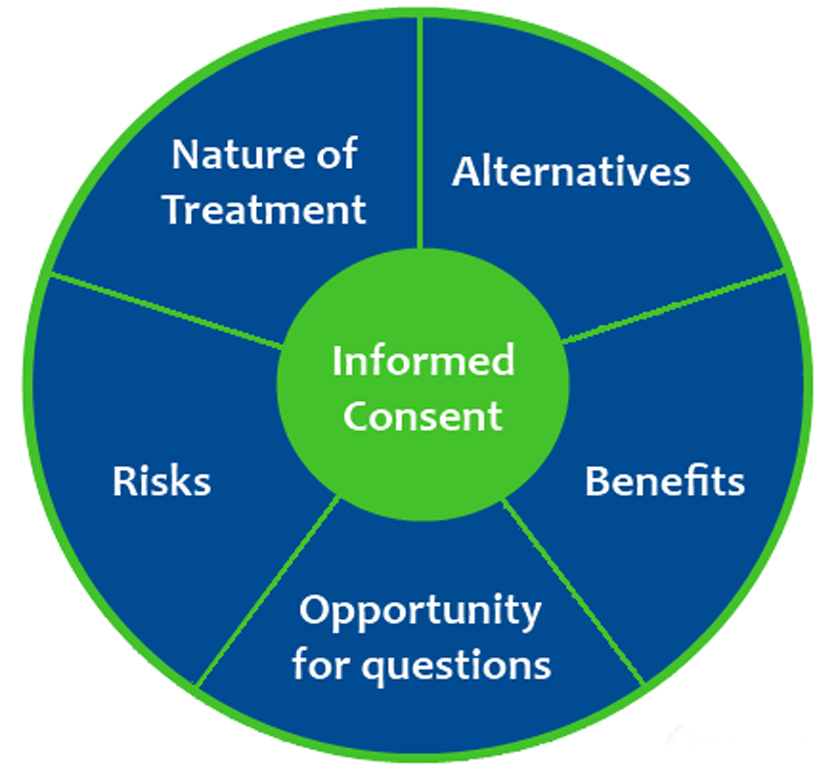
Informed Consent A Focus On Informed Myositis Support And Informed consent is a formal conversation between you and your healthcare provider about your treatment plan. it’s required by law, as well as by the medical code of ethics. By ‘informed consent’, we mean the process of providing patients with sufficient information to enable them to make a voluntary and informed decision regarding whether to undergo a procedure or not, and that the patients are able to understand the information given [4].

Understanding Informed Consent Informed consent is currently treated as the core of bioethics. in clinical practice, the doctrine of informed consent rose to dominance during the course of the 20th century. Informed consent (ic) is not just a signature or a document. ensures patients have enough information to make an informed decision about participating in clinical research. Informed consent is a fundamental component of ethical research involving human participants. it ensures that participants are fully aware of the nature of a study, its potential risks, benefits, and their right to withdraw at any time. at luth research, we prioritize informed consent as part of our commitment to ethical research practices and data. This process of understanding the risks and benefits of treatment is known as informed consent. informed consent is based on the moral and legal premise of patient autonomy: you as the patient have the right to make decisions about your own health and medical conditions.

Understanding Of Informed Consent Download Scientific Diagram Informed consent is a fundamental component of ethical research involving human participants. it ensures that participants are fully aware of the nature of a study, its potential risks, benefits, and their right to withdraw at any time. at luth research, we prioritize informed consent as part of our commitment to ethical research practices and data. This process of understanding the risks and benefits of treatment is known as informed consent. informed consent is based on the moral and legal premise of patient autonomy: you as the patient have the right to make decisions about your own health and medical conditions. Informed consent is when your healthcare provider gives you the information you need to make a decision about your care. you or a representative may need to formally acknowledge that you agree to certain procedures. In clinical research, informed consent is more than just a procedural requirement—it is the ethical foundation of participant engagement. it ensures individuals understand the purpose, risks, and benefits of a study before agreeing to participate. Informed consent in research informed consent requirements are especially rigorous in clinical research. federal regulations require researchers to disclose the purpose of the study, foreseeable risks and benefits, alternatives to participation, confidentiality protections, compensation if applicable, and contact information for questions. Before performing any invasive test or providing medical treatment, doctors must obtain permission from a competent patient in a manner that is informed and voluntary. the process is known as informed consent. (see also overview of legal and ethical issues in health care.).
Understanding Informed Patient Consent By Dr Bidushi Ganguli Collectlo Informed consent is when your healthcare provider gives you the information you need to make a decision about your care. you or a representative may need to formally acknowledge that you agree to certain procedures. In clinical research, informed consent is more than just a procedural requirement—it is the ethical foundation of participant engagement. it ensures individuals understand the purpose, risks, and benefits of a study before agreeing to participate. Informed consent in research informed consent requirements are especially rigorous in clinical research. federal regulations require researchers to disclose the purpose of the study, foreseeable risks and benefits, alternatives to participation, confidentiality protections, compensation if applicable, and contact information for questions. Before performing any invasive test or providing medical treatment, doctors must obtain permission from a competent patient in a manner that is informed and voluntary. the process is known as informed consent. (see also overview of legal and ethical issues in health care.).

Understanding The Informed Consent Process Docslib Informed consent in research informed consent requirements are especially rigorous in clinical research. federal regulations require researchers to disclose the purpose of the study, foreseeable risks and benefits, alternatives to participation, confidentiality protections, compensation if applicable, and contact information for questions. Before performing any invasive test or providing medical treatment, doctors must obtain permission from a competent patient in a manner that is informed and voluntary. the process is known as informed consent. (see also overview of legal and ethical issues in health care.).

Informed Consent
Comments are closed.