Understanding Design Controls

Design Controls Pdf Verification And Validation Risk Management Design control is a structured process that ensures your medical device does what it’s supposed to do while meeting safety and regulatory standards. the fda set up this framework to help developers follow high quality standards, reduce risks, and avoid errors before the product even hits the market. Design controls in medical devices are a systematic set of methods to carry out the design of medical devices. medical devices must be developed and manufactured in ways to ensure effectiveness, safety, and compliance.

Design Controls Process Flow Pdf Understand the importance of design controls in device quality and why they are a quality system requirement. know the quality system regulation requirements for design controls and learn. The document aims to assist medical device manufacturers in understanding and implementing effective design control practices to ensure the safety, performance, and overall quality of their. Design controls are a structured, systematic framework used in the engineering and development of complex or high risk products, such as medical devices. this disciplined approach ensures a product is built correctly and functions safely and effectively for its intended purpose. You can use this feature to further document your understanding of controls and to indicate controls that you plan to test. in general, you should focus on key controls.

Design Controls Digest 5 Phases Of Design Controls You Need To Know Design controls are a structured, systematic framework used in the engineering and development of complex or high risk products, such as medical devices. this disciplined approach ensures a product is built correctly and functions safely and effectively for its intended purpose. You can use this feature to further document your understanding of controls and to indicate controls that you plan to test. in general, you should focus on key controls. Design controls is a systematic method of developing a product that can potentially maximize the probability of making the “right product” to meet the user’s needs while minimizing the probability of exposing the user to harm. Understand medical device design controls with detailed insights into fda iso requirements, design inputs and outputs, flow charts, and examples for successful product development and approval. Design controls are a set of quality practices and procedures that are incorporated into the product design and development process to ensure that a device is appropriate for its intended use. Design controls are the application of a formal methodology to the conduct of product development activities. it is often mandatory (by regulation) to implement such practice when designing and developing products within regulated industries (e.g. medical devices).

Lecture 3 Design Controls And Criteria Pdf Lane Visual Perception Design controls is a systematic method of developing a product that can potentially maximize the probability of making the “right product” to meet the user’s needs while minimizing the probability of exposing the user to harm. Understand medical device design controls with detailed insights into fda iso requirements, design inputs and outputs, flow charts, and examples for successful product development and approval. Design controls are a set of quality practices and procedures that are incorporated into the product design and development process to ensure that a device is appropriate for its intended use. Design controls are the application of a formal methodology to the conduct of product development activities. it is often mandatory (by regulation) to implement such practice when designing and developing products within regulated industries (e.g. medical devices).

Design Controls For Medical Devices What You Need To Know Design controls are a set of quality practices and procedures that are incorporated into the product design and development process to ensure that a device is appropriate for its intended use. Design controls are the application of a formal methodology to the conduct of product development activities. it is often mandatory (by regulation) to implement such practice when designing and developing products within regulated industries (e.g. medical devices).
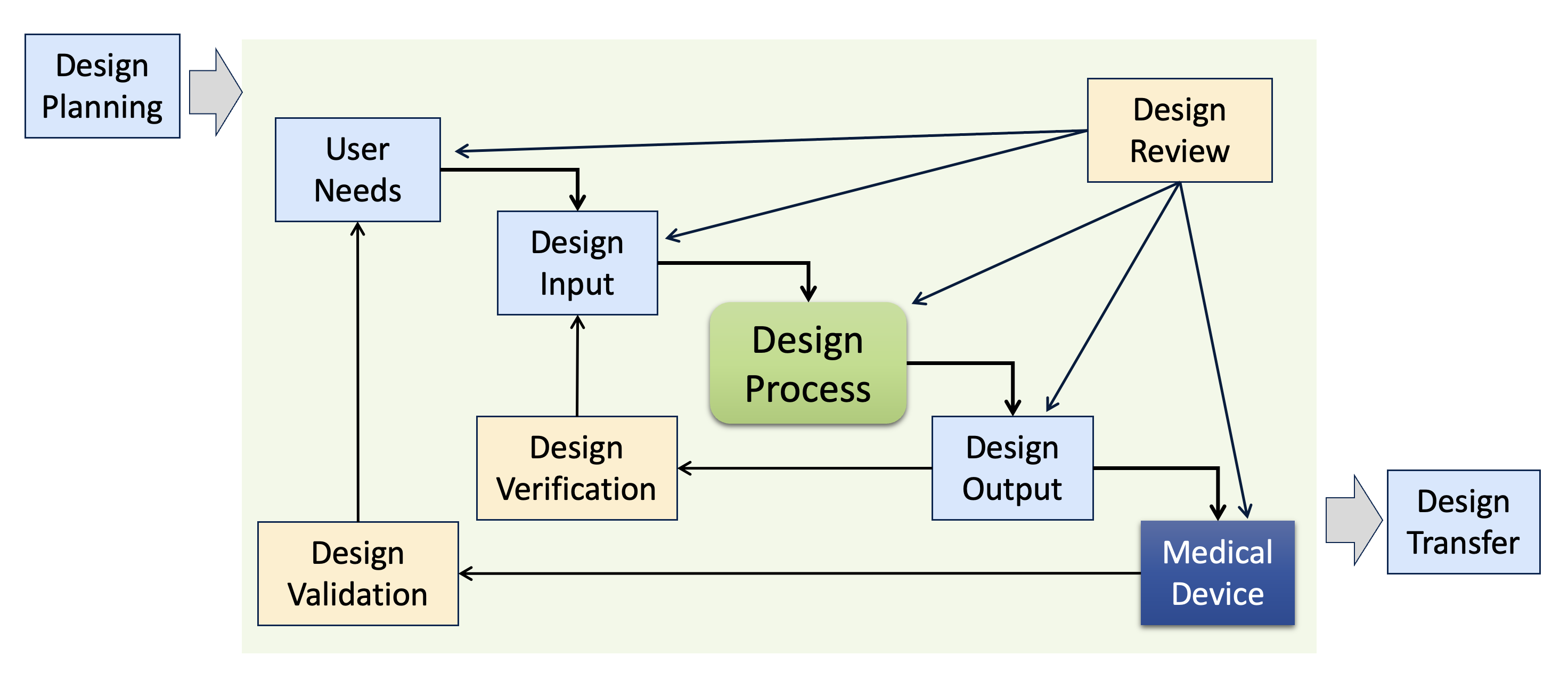
Design Controls Classic Diagram Sunstone Pilot Inc
Comments are closed.