Traceability In Medical Devices
Serialization And Traceability Of Medical Devices Arca Learn about iso 13485 traceability requirements, the benefits of identification systems, and implementation strategies for medical devices. This article outlines why traceability matters, the core elements of a strong system, and how medical device manufacturers can design and implement traceability in a practical, scalable way.

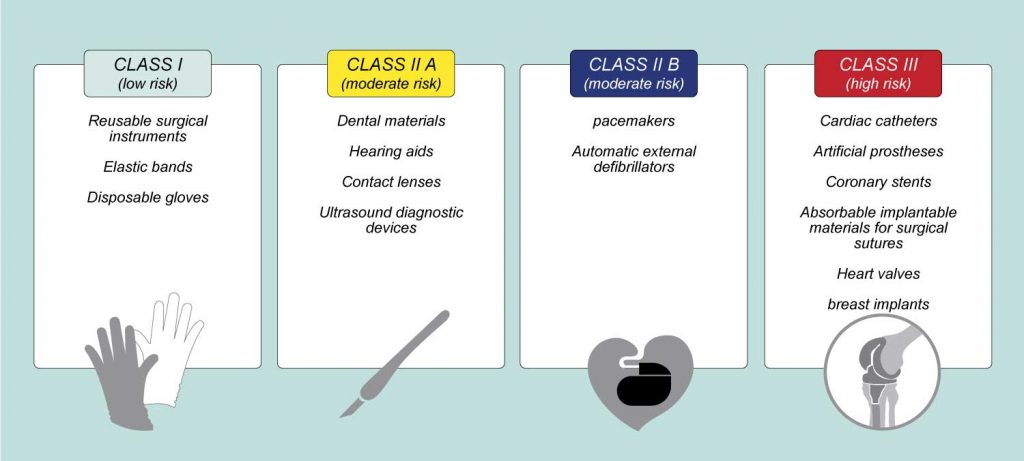
Traceability In Medical Devices What is medical device traceability? it is a function that tracks and traces a medical device from the site of initial manufacture to the site of patient or healthcare provider use. What is medical device traceability? medical device traceability is the systematic process of documenting, tracking, and tracing a medical device’s history, location, and usage throughout its entire lifecycle, from raw material sourcing and production to distribution and post market use. This article delves into the significance of the traceability matrix for medical devices, outlining the requirements, best practices, and the role they play in achieving regulatory approval. What is medical device traceability? medical device traceability ensures that medical devices and the materials they’re manufactured with can be tracked from prototyping and manufacturing to end use in a healthcare setting.

Udi The Traceability System For Medical Devices This article delves into the significance of the traceability matrix for medical devices, outlining the requirements, best practices, and the role they play in achieving regulatory approval. What is medical device traceability? medical device traceability ensures that medical devices and the materials they’re manufactured with can be tracked from prototyping and manufacturing to end use in a healthcare setting. However, when faced with the rigorous medical electronics field, the lack of iso 13485 certification means such general purpose manufacturers cannot provide the regulatory compliant traceability and quality management systems required, posing significant risks to the final device's compliance audit. Iso 13485 traceability is the ability to identify and track a medical device and its components, materials, and records throughout its entire lifecycle from incoming materials, production, distribution, and in some cases, to the final user or patient. In medical device development, traceability is far more than a formality. it is a regulatory requirement, a quality system cornerstone, and essential for ensuring safety and efficacy. the requirements traceability matrix (rtm) plays a vital role in meeting these expectations. Traceability is a key component of iso 13485 that involves tracking each device throughout its lifecycle, from raw material sourcing to production, distribution, and post market surveillance.
Comments are closed.