Tips For Writing Consent Forms Data Org

Tips For Writing Consent Forms Data Org Readability level guidelines for informed consent documents: the 2015 iom informed consent and health literacy workshop summary recommended that informed consent documents be written at the 8th grade reading level or lower. Use this checklist to help develop and or review consent forms. identify and know your potential participants. who are your potential participants? how familiar would you expect them to be with the study topic or research in general? consider potential participants’ perspectives.

Annex B Data Privacy Consent Form Updated Pdf Writing a research consent form means more than listing risks — here's what federal regulations actually require and where researchers often go wrong. Evaluate the record to ensure the data is findable, accessible, interoperable, and reusable in terms of the consent form, this means that the consent process is documented either internally as part of the repository’s metadata record or publicly as part of the readme file. Consent forms contain quite a lot of information. it’s good practice to allow your research subjects plenty of time to read and sign it. if you send the form to them via email and they reply, that implies consent. you’ll need to save and store the email in a way that makes it easy to track. The written presentation of information is used to document the basis for consent and for the participant’s future reference. the informed consent document should be revised when deficiencies are noted or when additional information will improve the consent process.
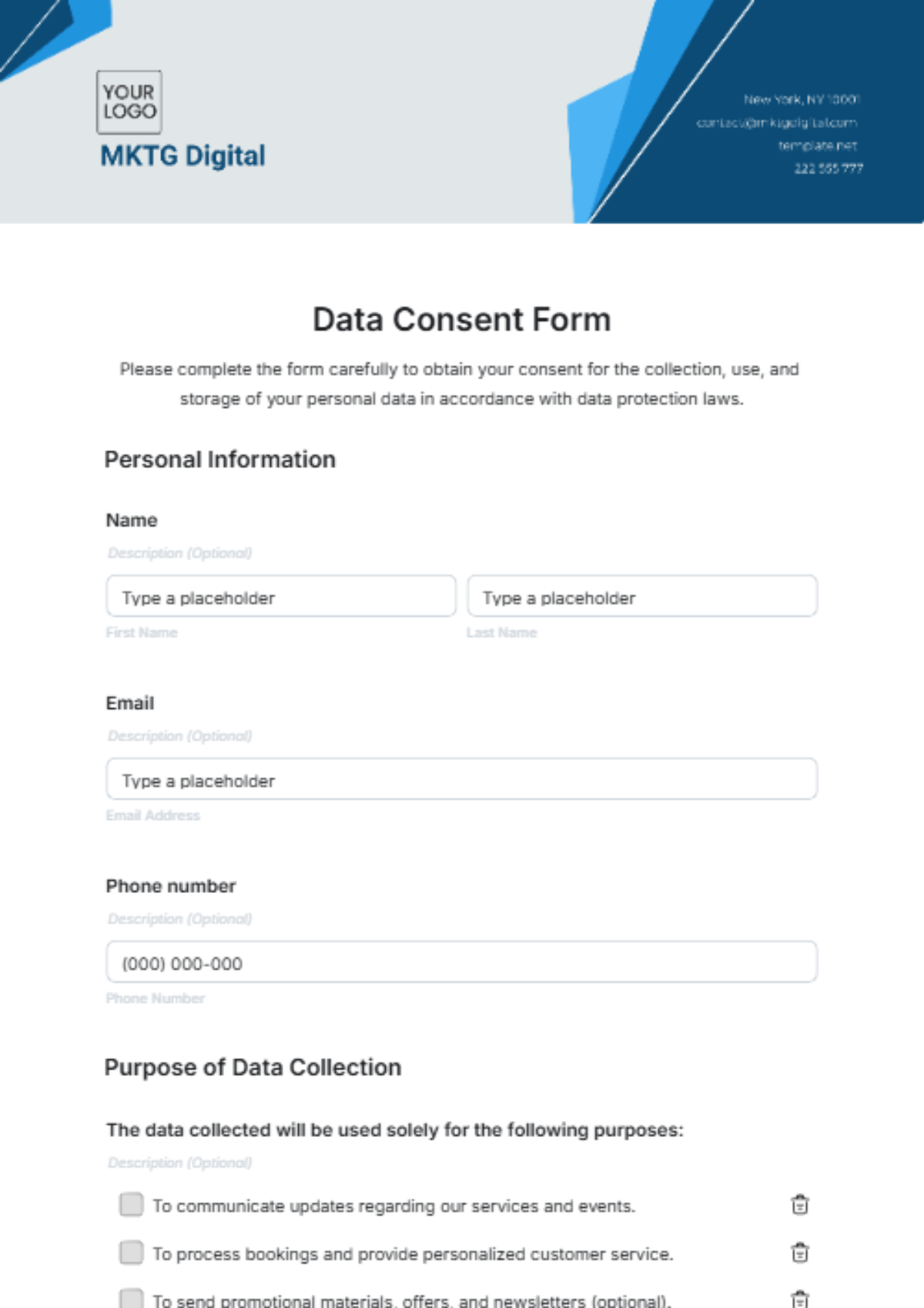
Free Consent Form Templates To Edit Online Consent forms contain quite a lot of information. it’s good practice to allow your research subjects plenty of time to read and sign it. if you send the form to them via email and they reply, that implies consent. you’ll need to save and store the email in a way that makes it easy to track. The written presentation of information is used to document the basis for consent and for the participant’s future reference. the informed consent document should be revised when deficiencies are noted or when additional information will improve the consent process. The guidelines (to be used in conjunction with the cto sample consent form tem plate) below outline some points that may be considered when drafting a consent form. If you prefer, consent forms can be written in the language which the researcher is most comfortable with and translated into the participants' mother tongue and english. ideally, translated consent forms should be back translated and compared with the original to ensure accuracy. Different methods include return of a signed form, returning an anonymous survey (providing tacit consent), and audio recording of verbal consent. other means may be appropriate depending on the nature of the research. A series of templates developed by the who erc to assist researchers in the design of their informed consent forms. access resource who.int rpc research ethics informed consent en this document is a suggested additional resource in our responsible data guidelines.

Personal Data Consent Form Pdpa Consent Template At Jayden Nobbs Blog The guidelines (to be used in conjunction with the cto sample consent form tem plate) below outline some points that may be considered when drafting a consent form. If you prefer, consent forms can be written in the language which the researcher is most comfortable with and translated into the participants' mother tongue and english. ideally, translated consent forms should be back translated and compared with the original to ensure accuracy. Different methods include return of a signed form, returning an anonymous survey (providing tacit consent), and audio recording of verbal consent. other means may be appropriate depending on the nature of the research. A series of templates developed by the who erc to assist researchers in the design of their informed consent forms. access resource who.int rpc research ethics informed consent en this document is a suggested additional resource in our responsible data guidelines.

Tips For Writing Consent Forms Data Org Different methods include return of a signed form, returning an anonymous survey (providing tacit consent), and audio recording of verbal consent. other means may be appropriate depending on the nature of the research. A series of templates developed by the who erc to assist researchers in the design of their informed consent forms. access resource who.int rpc research ethics informed consent en this document is a suggested additional resource in our responsible data guidelines.
Comments are closed.