Thermodynamics Closed System

Closed System Thermodynamics Lecture Notes Thermodynamics Docsity Definition: what is a closed system? a closed system is a fundamental concept in thermodynamics and physical sciences that describes an environment that does not exchange mass with its surroundings, but does allow the exchange of energy. A closed system can exchange energy with its surroundings through heat and work transfer. in other words, work and heat are the forms that energy can be transferred across the system boundary.
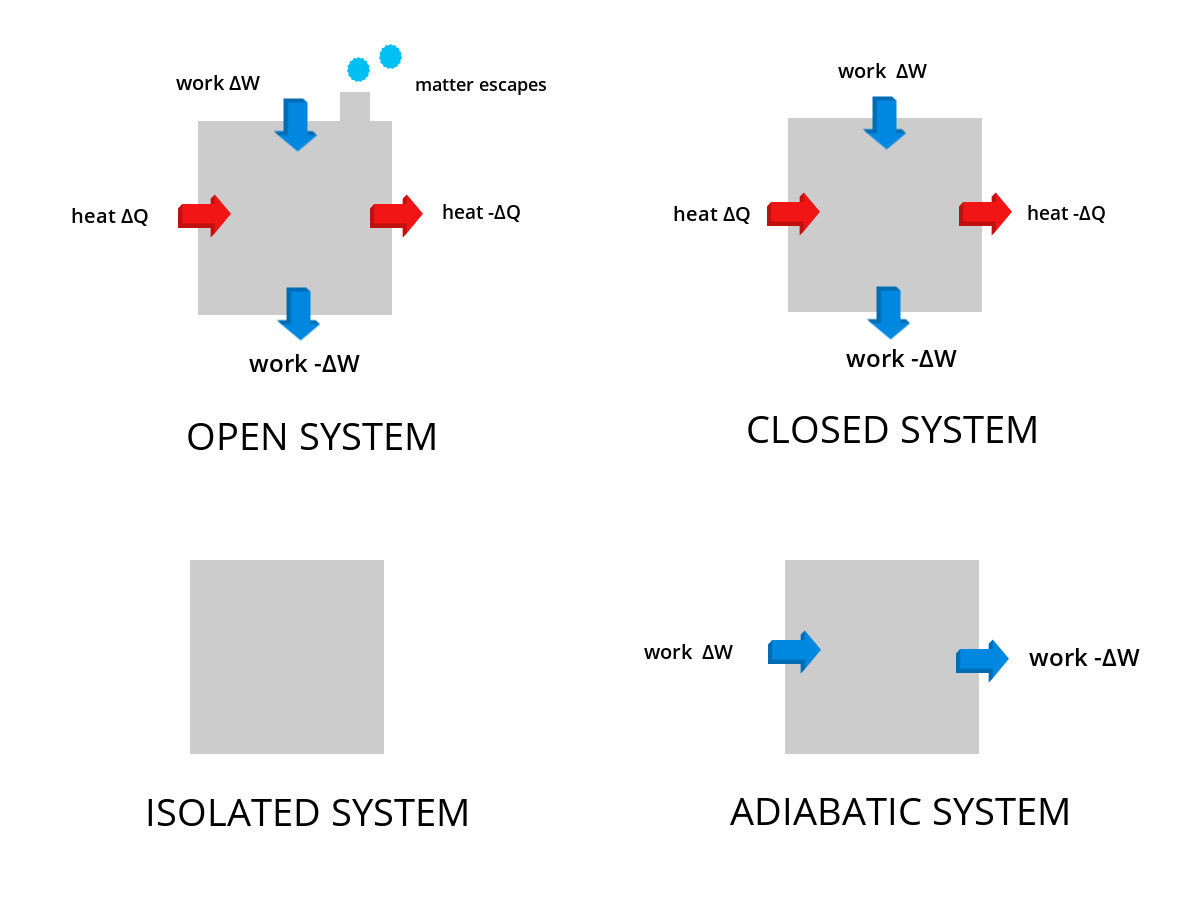
Themodynamic Systems Open Closed Isolated Systems Energy analysis of closed systems in this chapter, we apply the energy balance relation to systems that do not involve any mass flow across their boundaries; that is, closed systems. A closed system is a type of thermodynamic system that can exchange heat with its surroundings but mass cannot be exchanged. a pressure cooker is an example of a closed system. In thermodynamics, a closed system can exchange energy (as heat or work) but not matter, with its surroundings. Thermodynamics cannot tell about the rate (kinetics) of a process, but it can tell whether or not it is possible for a process to occur. for this, we make use of the first and second laws of thermodynamics.
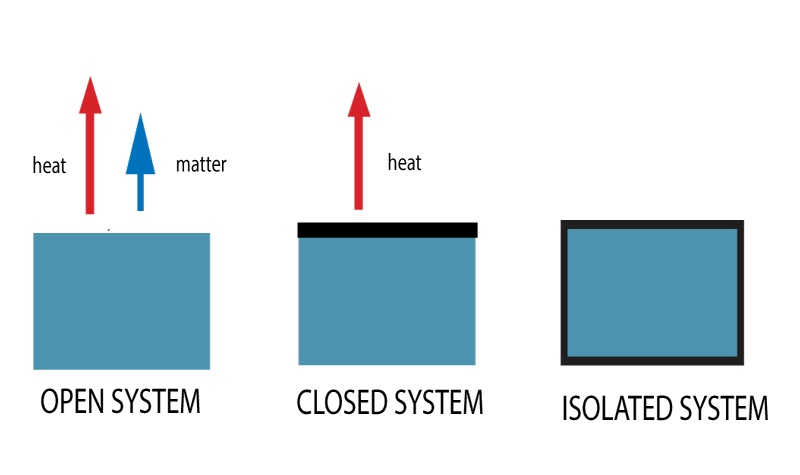
Themodynamic Systems Open Closed Isolated Systems In thermodynamics, a closed system can exchange energy (as heat or work) but not matter, with its surroundings. Thermodynamics cannot tell about the rate (kinetics) of a process, but it can tell whether or not it is possible for a process to occur. for this, we make use of the first and second laws of thermodynamics. For a closed system (no mass transfer) process proceeding between two states: Δ e = Δ k e Δ p e Δ u = q w. this is one to commit to memory! energy is transferred between the system and the surroundings in the form of heat and work, resulting in a change of total energy of the system. In thermodynamics, a closed system exchanges energy with its surroundings but does not exchange mass. this restriction allows engineers to concentrate on energy transformations—such as heating, cooling, compression, and expansion—making closed systems fundamental in many mechanical applications. Energy balance for closed systems is a crucial concept in thermodynamics. it applies the first law to systems that don't exchange matter with their surroundings, focusing on energy transfers through heat and work. understanding this topic helps you analyze various processes in closed systems. A thermodynamic system is classified as a closed energy system when it exchanges energy with its surroundings solely in the forms of thermal energy and work while its mass remains constant (i.e., no mass enters or leaves the system).

Closed System Cycles Thermodynamics Carnot Cycle Entropy Class 10 For a closed system (no mass transfer) process proceeding between two states: Δ e = Δ k e Δ p e Δ u = q w. this is one to commit to memory! energy is transferred between the system and the surroundings in the form of heat and work, resulting in a change of total energy of the system. In thermodynamics, a closed system exchanges energy with its surroundings but does not exchange mass. this restriction allows engineers to concentrate on energy transformations—such as heating, cooling, compression, and expansion—making closed systems fundamental in many mechanical applications. Energy balance for closed systems is a crucial concept in thermodynamics. it applies the first law to systems that don't exchange matter with their surroundings, focusing on energy transfers through heat and work. understanding this topic helps you analyze various processes in closed systems. A thermodynamic system is classified as a closed energy system when it exchanges energy with its surroundings solely in the forms of thermal energy and work while its mass remains constant (i.e., no mass enters or leaves the system).
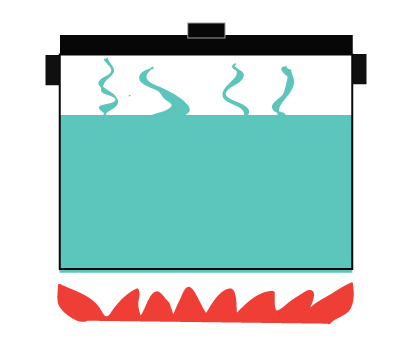
Themodynamic Systems Open Closed Isolated Systems Energy balance for closed systems is a crucial concept in thermodynamics. it applies the first law to systems that don't exchange matter with their surroundings, focusing on energy transfers through heat and work. understanding this topic helps you analyze various processes in closed systems. A thermodynamic system is classified as a closed energy system when it exchanges energy with its surroundings solely in the forms of thermal energy and work while its mass remains constant (i.e., no mass enters or leaves the system).
Comments are closed.