The Quantum Wavefunction Explained

The Quantum Wavefunction Explained Classx In quantum physics, a wave function (or wavefunction) is a mathematical description of the quantum state of an isolated quantum system. the most common symbols for a wave function are the greek letters ψ and Ψ (lower case and capital psi, respectively). The wavefunction is a foundational concept in quantum mechanics, providing a mathematical and conceptual framework for understanding the behavior of particles. by describing probabilities and superpositions, it explains everything from the structure of atoms to the behavior of quantum systems.

Supwerposition Quantum Illustarted And With Examples Quantum At its core, a wave function is a complex valued mathematical function, typically denoted by the greek letter ψ (psi). it describes the quantum state of a system — be it a single particle, like an electron, or an entire collection of particles. Wave function, in quantum mechanics, variable quantity that mathematically describes the wave characteristics of a particle. the value of the wave function of a particle at a given point of space and time is related to the likelihood of the particle’s being there at the time. Delve into the core of quantum mechanics with the wavefunction and schrödinger’s equation. discover how they describe particle probabilities, time evolution, and the behavior of quantum systems, forming the basis of modern physics. The lesson on “the quantum wavefunction explained” delves into the nature of quantum wave functions, which describe the behavior of particles in quantum mechanics through the schrödinger equation.
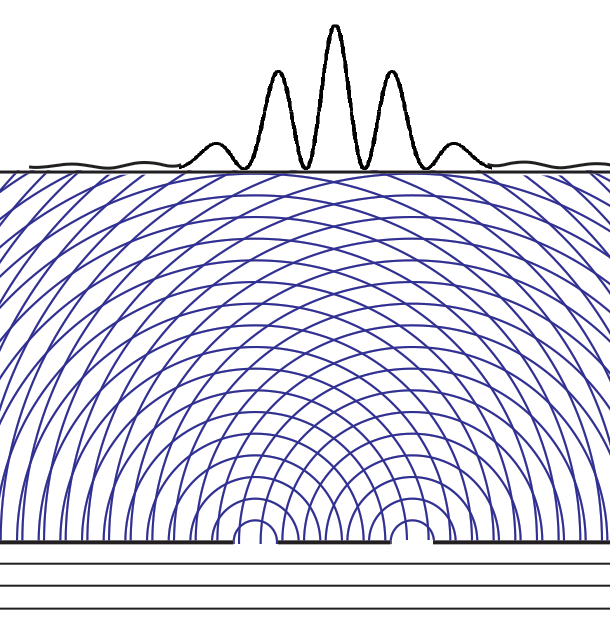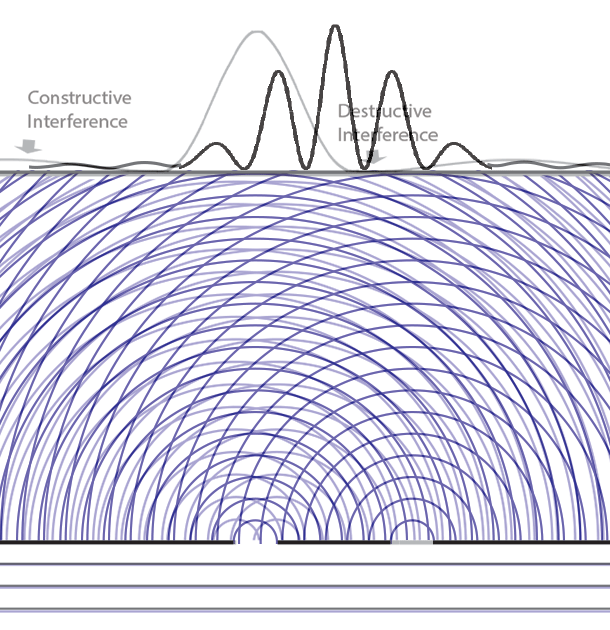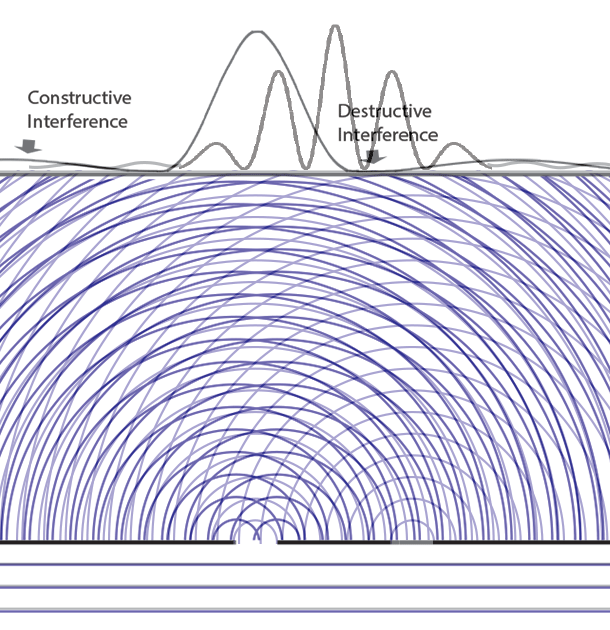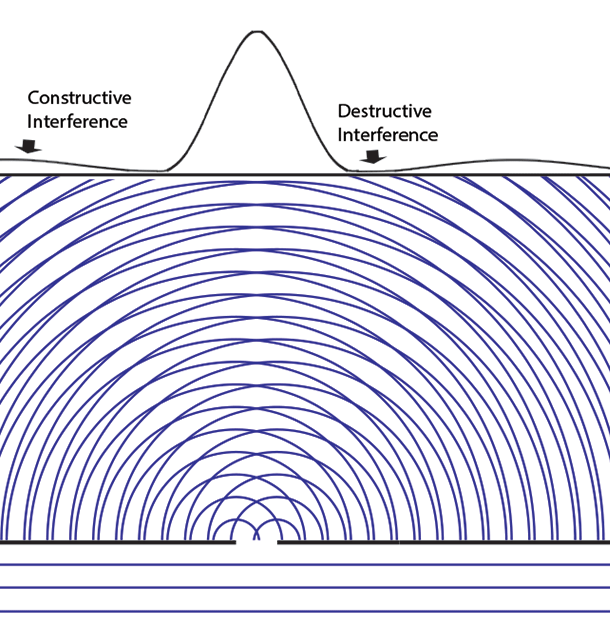
Collapse Of The Wave Function Delve into the core of quantum mechanics with the wavefunction and schrödinger’s equation. discover how they describe particle probabilities, time evolution, and the behavior of quantum systems, forming the basis of modern physics. The lesson on “the quantum wavefunction explained” delves into the nature of quantum wave functions, which describe the behavior of particles in quantum mechanics through the schrödinger equation. Given two possible states of a quantum system corresponding to two wavefunctions ψa and ψb, the system could also be in a superposition ψ = αψa βψb with α and β as arbitrary complex coefficients satisfying normalization. The behavior of quantum particles, such as electrons and photons, is governed by the evolution of their wave functions over time, as described by schrödinger's equation. Understand the concept of wave function in quantum mechanics, including its physical meaning, mathematical form, and applications. simplified for students. In quantum mechanics, however, the solution to an equation of motion is a wavefunction, Ψ (x, t). the particle has many values of position for any time t, and only the probability density of finding the particle, | Ψ (x, t) | 2, can be known.
Comments are closed.