The Heisenberg Uncertainty Principle Part 1 Position Momentum And Schrodingers Cat

Heisenberg Uncertainty Principle Scienly He has a few things to say about what we can and can't know about the quantum world. then we will wrap things up with the most famous cat in the world after garfield, schrödinger's cat. It states that there is a limit to the precision with which certain pairs of physical properties, such as position and momentum, can be simultaneously known. in other words, the more accurately one property is measured, the less accurately the other property can be known.

Heisenberg Uncertainty Principle And Its Impact Cd Sears Use both versions of heisenberg’s uncertainty principle in calculations. explain the implications of heisenberg’s uncertainty principle for measurements. The heisenberg uncertainty principle part 1: position momentum and schrödinger's cat. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa. Does the uncertainty principle only apply to position and momentum? no, it applies to all conjugate variable pairs, including energy time and angular quantities.
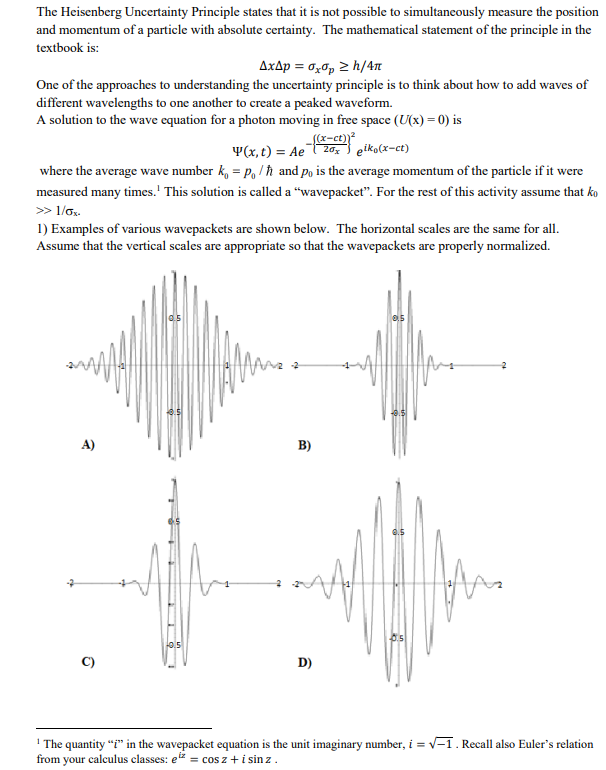
Solved The Heisenberg Uncertainty Principle States That It Chegg Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa. Does the uncertainty principle only apply to position and momentum? no, it applies to all conjugate variable pairs, including energy time and angular quantities. Heisenberg’s uncertainty principle is a key principle in quantum mechanics. very roughly, it states that if we know everything about where a particle is located (the uncertainty of position is small), we know nothing about its momentum (the uncertainty of momentum is large), and vice versa. According to quantum mechanics, the more precisely the position (momentum) of a particle is given, the less precisely can one say what its momentum (position) is. this is (a simplistic and preliminary formulation of) the quantum mechanical uncertainty principle for position and momentum. One of the bedrock tenets of quantum physics is the uncertainty principle, formulated by werner heisenberg in the 1920s. it says that certain pairs of quantum properties—such as position (x) and momentum (p) always maintain a careful balance. Measuring devices are essentially classical devices, and measure only classical properties such as position and momentum. the quantum mechanical description of large systems will closely approximate the classical description.
Comments are closed.