The Easiest Rule For Chemical Reactions The 7 Days Chemistry Challenge Day 5 Delowar Sir %f0%9f%92%a5
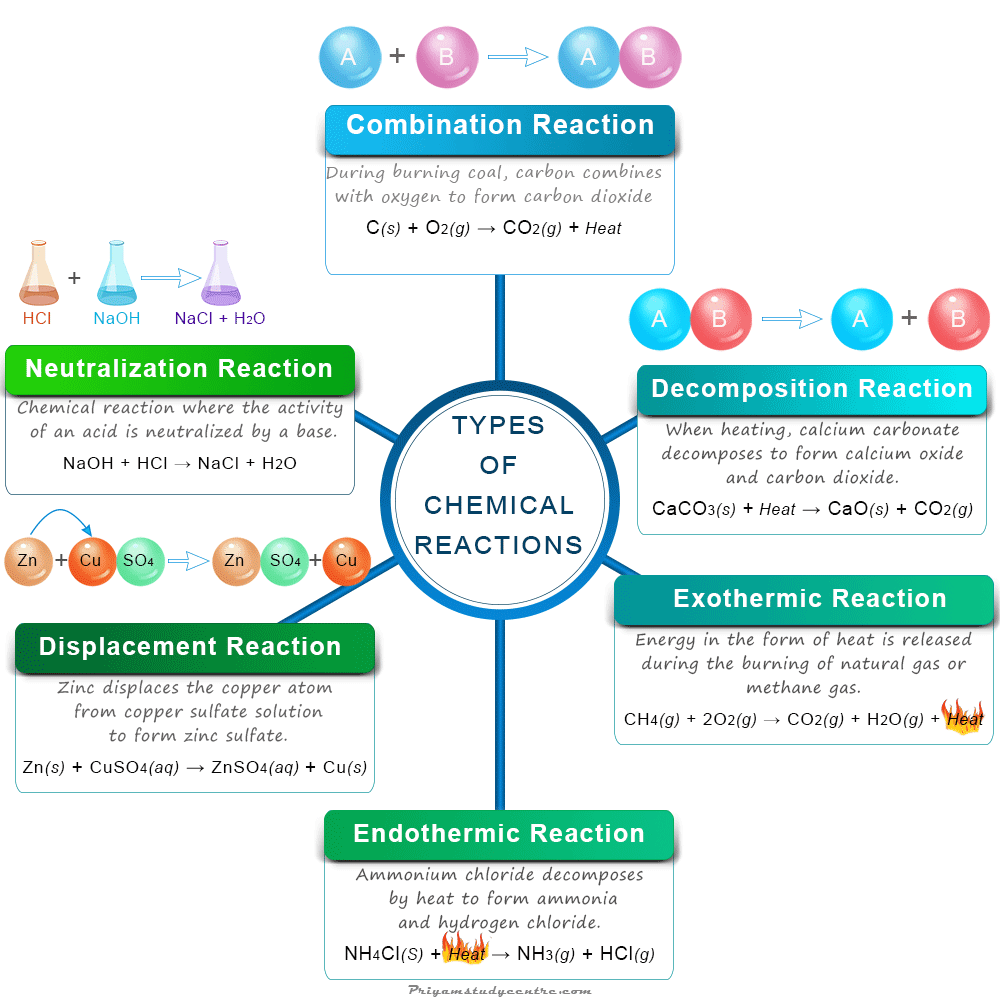
Chemical Reactions Examples So now it’s time to conquer chemistry without being afraid of it!. Zero to hero in chemistry in 7 days | 7 days chemistry challenge | delowar sir unique teaching method • 9.2k views • 4 months ago.

Types Of Reactions Worksheet Resource Twinkl Usa Delwar sir brings you "the 7 days chemistry challenge" 7 days topics: 📘 day 1 — the birth story of the yojani 📗 day 2 — the story of metals and non metals 📕 day 3 — the myth of the. During chemical reactions or a change of state, no atoms are created or destroyed. the total mass of chemicals before and after a reaction remains the same. what is combustion? combustion. Things that are mandatory to know in chemistry. the videos in this playlist will overcome the fear of chemistry and create a new love for chemistry in you, insha allah. This page covers chemical reactions, explaining how substances transform into new ones with altered properties through the breaking of bonds and rearrangement of atoms. it includes examples like rusting and combustion and discusses observable signs of reactions such as color changes and gas release.

Preset Alight F0 9f 98 88 F0 9f 92 A5 Motion Ff F0 9f 92 A5 F0 9f 8e B6 Things that are mandatory to know in chemistry. the videos in this playlist will overcome the fear of chemistry and create a new love for chemistry in you, insha allah. This page covers chemical reactions, explaining how substances transform into new ones with altered properties through the breaking of bonds and rearrangement of atoms. it includes examples like rusting and combustion and discusses observable signs of reactions such as color changes and gas release. Unlock the world of chemical reactions! get ready to decode the language of chemistry, using symbols and equations to gain a deeper understanding of how substances interact and change at the atomic level. Use these free chemistry lesson plans, experiments, and activities to teach and explore chemical reactions with k 12 students. a chemical reaction occurs when two or more substances (reactants) are mixed together and result in one or more new substances (products). Stoichiometry: relationship of reactants and products in a chemical reaction, based on mass (g) and or moles (number of molecules). always relates two different substances via their mole ratios. “mass is neither created nor destroyed in chemical reactions.” in other words, and for a closed system, the total mass of both the reactants and the products of a chemical reaction will remain the same throughout that reaction and at any point in time during that reaction.

Ssc Chemistry Chapter 5 а ёа а ќа а їа їа ј а їа а ња ёа ђ а ёа ѓа єа ќа а їа а ња ёа ђ Delowar Sir Unlock the world of chemical reactions! get ready to decode the language of chemistry, using symbols and equations to gain a deeper understanding of how substances interact and change at the atomic level. Use these free chemistry lesson plans, experiments, and activities to teach and explore chemical reactions with k 12 students. a chemical reaction occurs when two or more substances (reactants) are mixed together and result in one or more new substances (products). Stoichiometry: relationship of reactants and products in a chemical reaction, based on mass (g) and or moles (number of molecules). always relates two different substances via their mole ratios. “mass is neither created nor destroyed in chemical reactions.” in other words, and for a closed system, the total mass of both the reactants and the products of a chemical reaction will remain the same throughout that reaction and at any point in time during that reaction.

Preset Alight F0 9f 91 Bf F0 9f 92 A5 Motion Ff F0 9f 92 A5 F0 9f 8e Stoichiometry: relationship of reactants and products in a chemical reaction, based on mass (g) and or moles (number of molecules). always relates two different substances via their mole ratios. “mass is neither created nor destroyed in chemical reactions.” in other words, and for a closed system, the total mass of both the reactants and the products of a chemical reaction will remain the same throughout that reaction and at any point in time during that reaction.
Comments are closed.