Synthesis Reactions

Chemical Reactions Synthesis And Decomposition Worksheet Live A synthesis reaction is a chemical reaction that combines two or more simple elements or compounds to form a more complex product. learn how to identify a synthesis reaction, see examples of different types of synthesis reactions, and compare them with other reaction types. A synthesis reaction is a reaction in which two or more reactants chemically bond and combine to form a product. learn how to identify a synthesis reaction, see different types of synthesis reactions, and find examples of synthesis reactions in everyday life.

12 Reaction Types Worksheet Key Pdf Worksheets Library In a synthesis reaction, two or more substances combine to form a single, more complex substance. a synthesis reaction is sometimes referred to as a combination reaction. A synthesis reaction is defined as a chemical process in which reactants combine to form a more complex product, exemplified by the catalytic conversion of syngas (carbon monoxide and hydrogen) into methanol through an exothermic reaction. Learn what synthesis reactions are, how they occur, and why they are important in chemistry and industry. find examples of synthesis reactions in inorganic and organic chemistry, and how to balance chemical equations for them. A synthesis reaction or direct combination reaction is a type of chemical reaction in which two or more simple substances combine to form a more complex product.
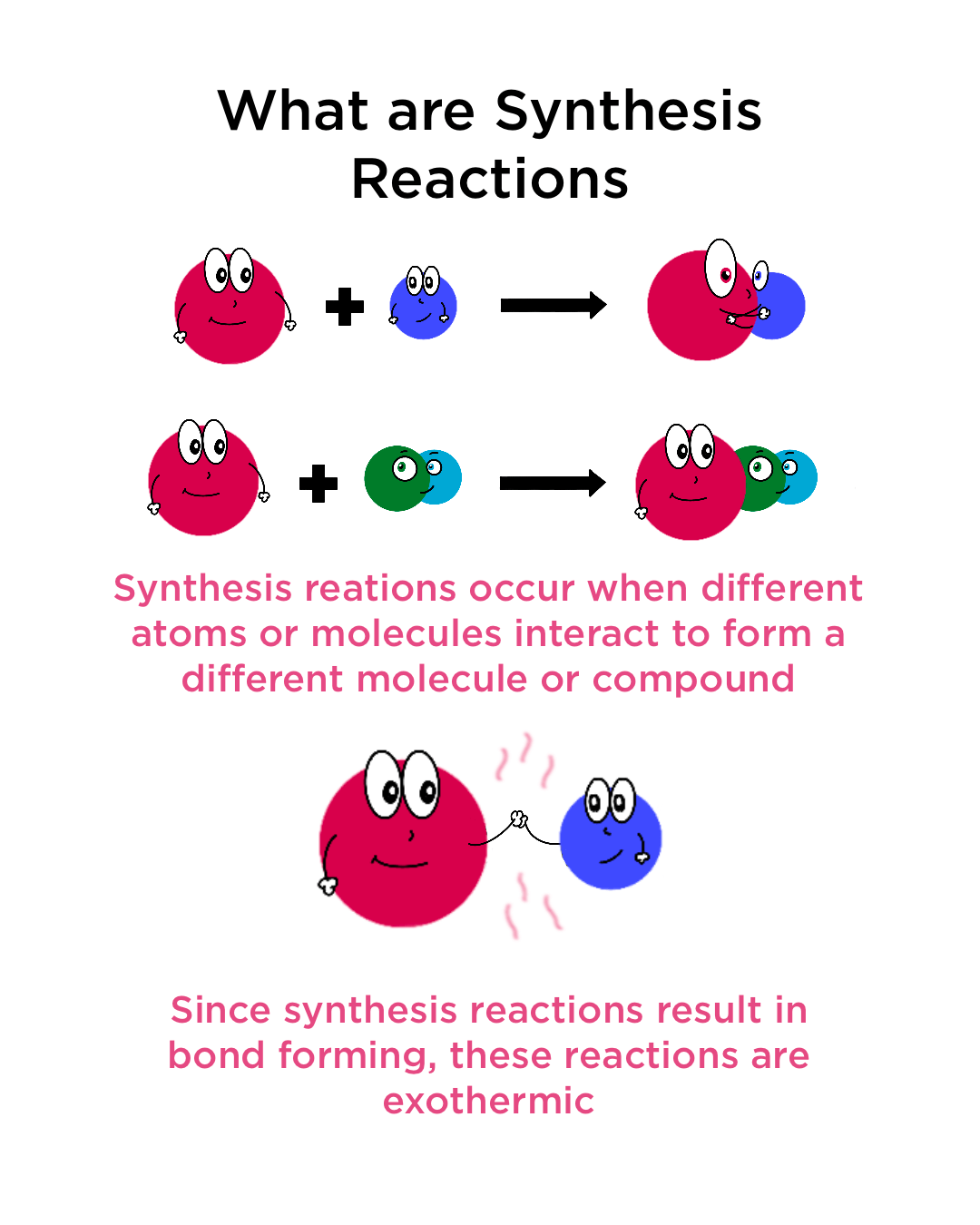
Synthesis Reactions Definition Examples Expii Learn what synthesis reactions are, how they occur, and why they are important in chemistry and industry. find examples of synthesis reactions in inorganic and organic chemistry, and how to balance chemical equations for them. A synthesis reaction or direct combination reaction is a type of chemical reaction in which two or more simple substances combine to form a more complex product. Synthesis reactions, fundamental to chemical engineering and material science, represent a core class of chemical transformations where two or more chemical species (atoms, ions, or molecules) combine to form a more complex product. A synthesis reaction, also known as a combination reaction, occurs when two or more simpler substances combine to form a single, more complex product. this type of reaction can involve elements forming a compound, or simpler compounds combining to form a more intricate one. Learn about synthesis reactions in chemistry: definition, examples, and facts. includes interactive quiz on chemical reactions, reactants, products, and bonding. Synthesis reactions, also known as combination reactions, are a type of chemical reaction where two or more reactants combine to form a single product. these reactions result in the creation of a new substance with different properties than the original reactants.
Comments are closed.