Synthesis Of 35 Dimethylpyrazole

Solved Propose A Synthesis And Mechanism To Account For 3 5 The document describes the synthesis of 3,5 dimethylpyrazole through the reaction of acetylacetone and hydrazine hydrate. the reaction yields a crystalline solid that was filtered and dried. Synthesis of 3,5 dimethylpyrazole from acetylacetone.chemicalbook can provide 10 synthetic routes.

Scheme 30 Mechanism For The Synthesis Of 3 5 2 Hydroxyaryl Pyrazoles The following diagram illustrates the main reaction pathway for the synthesis of 3,5 dimethylpyrazole from acetylacetone and hydrazine, along with a potential side reaction leading to the formation of acetylacetone azine. In this video i follow the procedure in vogel's practical organic chemistry on the synthesis of 3,5 dimethylpyrazole by the condensation of hydrazine with acetylacetone. Synthesis of 3 5 dimethylpyrazole [j3noq8v1e54d]. The ether extracts are combined, washed once with saturated sodium chloride solution, and dried over anhydrous potassium carbonate. the ether is removed by distillation, and the slightly yellow residue of crystalline 3,5 dimethylpyrazole obtained by drying at reduced pressure (approximately 20 mm.) weighs 37–39 g. (77–81%), m.p. 107–108°.

Pdf Synthesis Of N Vinylformamide And 1 Vinyl 1 Methacryloyl 3 5 Synthesis of 3 5 dimethylpyrazole [j3noq8v1e54d]. The ether extracts are combined, washed once with saturated sodium chloride solution, and dried over anhydrous potassium carbonate. the ether is removed by distillation, and the slightly yellow residue of crystalline 3,5 dimethylpyrazole obtained by drying at reduced pressure (approximately 20 mm.) weighs 37–39 g. (77–81%), m.p. 107–108°. The laboratory synthesis of 3,5 dimethylpyrazole primarily involves the condensation of acetylacetone (pentane 2,4 dione) with hydrazine or a hydrazine salt under mild conditions. 3,5 dimethylpyrazole is an organic compound with the formula (ch 3 c) 2 chn 2 h. it is one of several isomeric derivatives of pyrazole that contain two methyl substituents. A deep dive into the chemical structure, synthesis pathways, and key properties of 3,5 dimethylpyrazole (cas 67 51 6), essential for chemists and material scientists. Rothenberg 1 synthesized 3,5 dimethylpyrazole by reacting hydrazine hydrate and acetylacetone in ethanol, but due to violent reactivity of hydrazine, rosengarten2 acquired the substance by reacting hydrazine sulfate and acetylacetone in aqueous alkali.

Synthesis Of 3 5 Dimethylpyrazole Pdf Chemical Reactions Chemical The laboratory synthesis of 3,5 dimethylpyrazole primarily involves the condensation of acetylacetone (pentane 2,4 dione) with hydrazine or a hydrazine salt under mild conditions. 3,5 dimethylpyrazole is an organic compound with the formula (ch 3 c) 2 chn 2 h. it is one of several isomeric derivatives of pyrazole that contain two methyl substituents. A deep dive into the chemical structure, synthesis pathways, and key properties of 3,5 dimethylpyrazole (cas 67 51 6), essential for chemists and material scientists. Rothenberg 1 synthesized 3,5 dimethylpyrazole by reacting hydrazine hydrate and acetylacetone in ethanol, but due to violent reactivity of hydrazine, rosengarten2 acquired the substance by reacting hydrazine sulfate and acetylacetone in aqueous alkali.
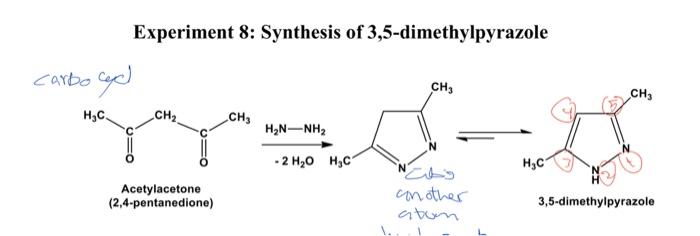
Solved Experiment 8 Synthesis Of 3 5 Dimethylpyrazole Chegg A deep dive into the chemical structure, synthesis pathways, and key properties of 3,5 dimethylpyrazole (cas 67 51 6), essential for chemists and material scientists. Rothenberg 1 synthesized 3,5 dimethylpyrazole by reacting hydrazine hydrate and acetylacetone in ethanol, but due to violent reactivity of hydrazine, rosengarten2 acquired the substance by reacting hydrazine sulfate and acetylacetone in aqueous alkali.

Pdf Synthesis Of 1 Ethylpyrazole 4 Carbaldehydes 1 1 Methylenebis 3
Comments are closed.