Super Common Mistake Diatomic Elements

Diatomic Super Mutating Dynamic Food Replicator Autosaved Pdf Many students get confused by the diatomic elements. bromine, iodine, nitrogen, chlorine, hydrogen, oxygen, and fluorine always form diatomic molecules and pair up. While only these seven elements routinely form diatomic molecules, other elements can form them. however, diatomic molecules formed by other elements are not very stable, so their bonds are easily broken.

What Are The 7 Diatomic Elements Definition And List There are seven diatomic elements: hydrogen, nitrogen, oxygen, fluorine, chlorine, iodine, bromine. video advice: super common mistake: diatomic elements. shouldn’t there be two atoms of every diatomic element? many students get confused by the diatomic elements. This is a list of all of the diatomic elements and their common properties. simple mnemonics for remembering them are included. A diatomic molecule is a molecule that has two atoms, but the atoms can be different. for example, carbon monoxide (co) is a diatomic molecule, but not a diatomic element. We are constantly referring to the elements on the periodic table. seven of those elements exist as diatomic molecules meaning the formula is not just the symbol by itself, but the symbol subscript 2.
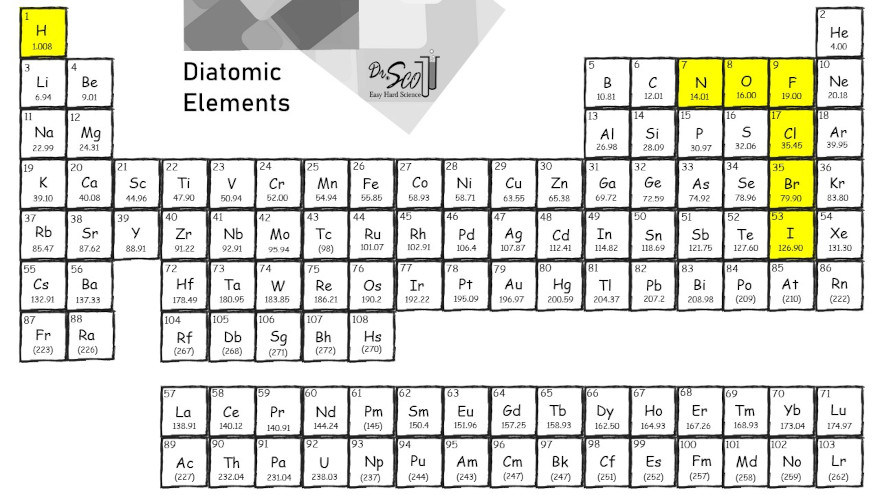
Diatomic Elements Easy Hard Science A diatomic molecule is a molecule that has two atoms, but the atoms can be different. for example, carbon monoxide (co) is a diatomic molecule, but not a diatomic element. We are constantly referring to the elements on the periodic table. seven of those elements exist as diatomic molecules meaning the formula is not just the symbol by itself, but the symbol subscript 2. Every molecule has its own unique molecular formula, and for diatomic elements, their formula always contains a subscript of 2, which represents two atoms in its structure. To remember the 7 diatomic elements (h, n, o, f, cl, br, i), use the mnemonic “have no fear of ice cold beer” or the element symbol based mnemonic “hofbrincl”. these memory tricks make recalling the names and symbols easy and fast whenever needed in chemistry. Diatomic elements hate to be alone — so much so that they just aren't found as single atoms. instead, they're always two atoms of the same pure element bonded together. While only these seven elements usually form diatomic molecules, additional elements can as well. however, diatomic molecules generated by other elements are not particularly stable, and their bonds are rapidly broken.

Diatomic Elements Teaching Resources Every molecule has its own unique molecular formula, and for diatomic elements, their formula always contains a subscript of 2, which represents two atoms in its structure. To remember the 7 diatomic elements (h, n, o, f, cl, br, i), use the mnemonic “have no fear of ice cold beer” or the element symbol based mnemonic “hofbrincl”. these memory tricks make recalling the names and symbols easy and fast whenever needed in chemistry. Diatomic elements hate to be alone — so much so that they just aren't found as single atoms. instead, they're always two atoms of the same pure element bonded together. While only these seven elements usually form diatomic molecules, additional elements can as well. however, diatomic molecules generated by other elements are not particularly stable, and their bonds are rapidly broken.
Comments are closed.