Submission Types
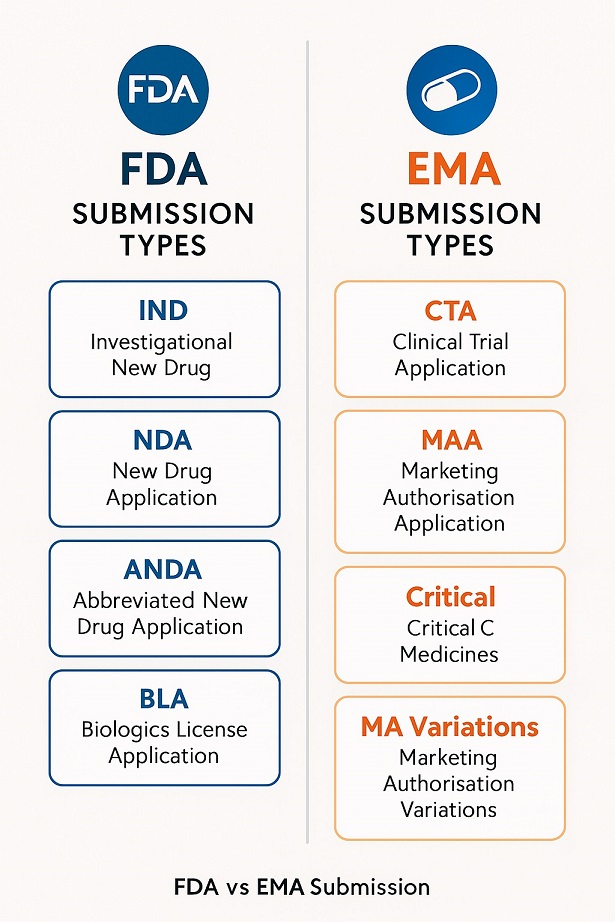
The Ultimate Guide To Regulatory Submissions There are several types of regulatory submissions that you’ll need to navigate throughout the life cycle of a drug. the two most significant types are clinical trial applications and marketing authorisation applications. Pharmaceutical companies must navigate different submission types throughout a product’s lifecycle. understanding these submissions is essential for efficient regulatory planning and compliance.
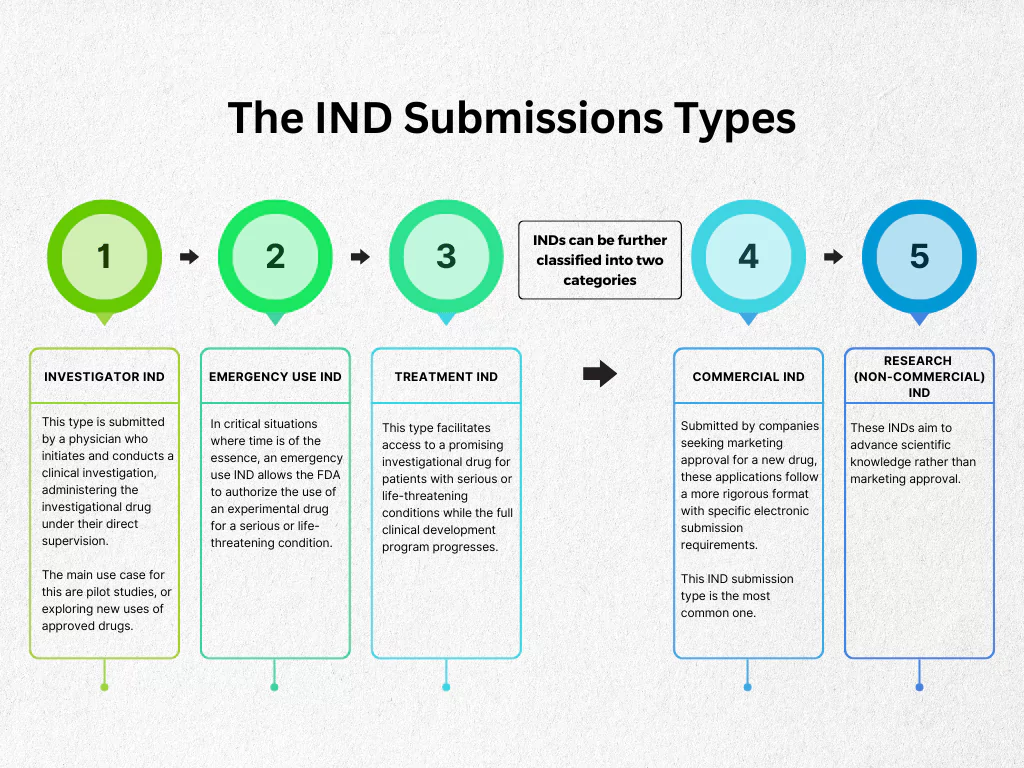
Investigational New Drug Ind Application Process An A Z Guide As the product reaches the market, companies must submit updates, monitor safety, and report changes. therefore, a strong regulatory strategy ensures compliance from start to finish. This blog talks about common types of regulatory submissions in pharma and their importance for global approvals. Various types of submissions exist, including ind, nda, cta, and maa, each with specific requirements and regions of application. these submissions are crucial for legal compliance and maintaining product quality throughout the development and post market phases. Regulatory submissions in the pharmaceutical industry are categorised by development stage, product type, and regulatory authority, including inds, ndas,.

Ppt Proper Irb Application Guidance For Research Projects Powerpoint Various types of submissions exist, including ind, nda, cta, and maa, each with specific requirements and regions of application. these submissions are crucial for legal compliance and maintaining product quality throughout the development and post market phases. Regulatory submissions in the pharmaceutical industry are categorised by development stage, product type, and regulatory authority, including inds, ndas,. Learn the regulatory submission process, key requirements for fda and eu approvals, and strategies to streamline submissions for faster market access. Depending on the stage of drug development, the intended market, and the product type, different regulatory documents are prepared and submitted. below is an overview of the key types of. Various types of regulatory submissions are essential in the pharmaceutical and medical device industries. these include investigational new drug (ind) applications for clinical trials, new drug applications (ndas) for new pharmaceuticals, biologics license applications (blas) for biological products, and premarket approval (pma) applications. To help you navigate the landscape, we offer this short guide to the most common regulatory submission types in the united states (fda) and european union (ema), with links to more detailed information.

Fda De Novo Pathway Guide And Best Practices Learn the regulatory submission process, key requirements for fda and eu approvals, and strategies to streamline submissions for faster market access. Depending on the stage of drug development, the intended market, and the product type, different regulatory documents are prepared and submitted. below is an overview of the key types of. Various types of regulatory submissions are essential in the pharmaceutical and medical device industries. these include investigational new drug (ind) applications for clinical trials, new drug applications (ndas) for new pharmaceuticals, biologics license applications (blas) for biological products, and premarket approval (pma) applications. To help you navigate the landscape, we offer this short guide to the most common regulatory submission types in the united states (fda) and european union (ema), with links to more detailed information.
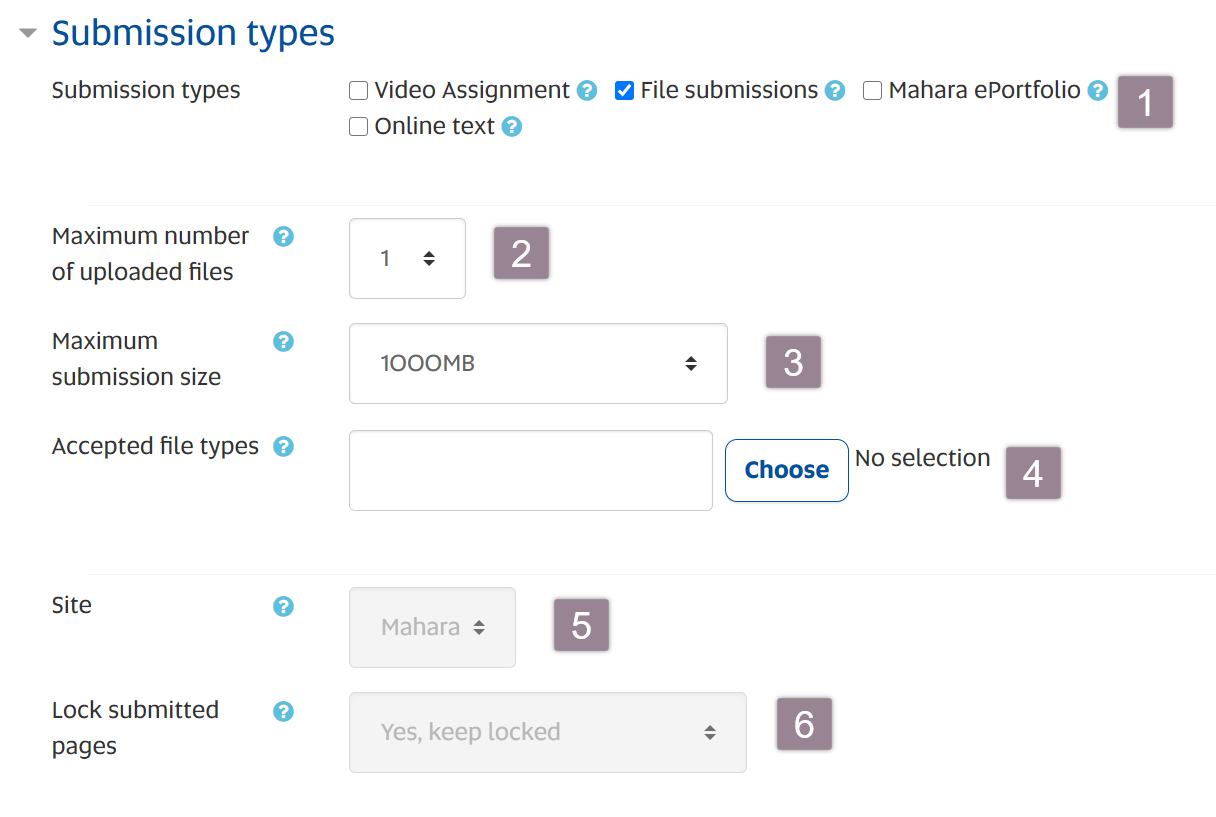
Assessment Settings Submission Types Various types of regulatory submissions are essential in the pharmaceutical and medical device industries. these include investigational new drug (ind) applications for clinical trials, new drug applications (ndas) for new pharmaceuticals, biologics license applications (blas) for biological products, and premarket approval (pma) applications. To help you navigate the landscape, we offer this short guide to the most common regulatory submission types in the united states (fda) and european union (ema), with links to more detailed information.
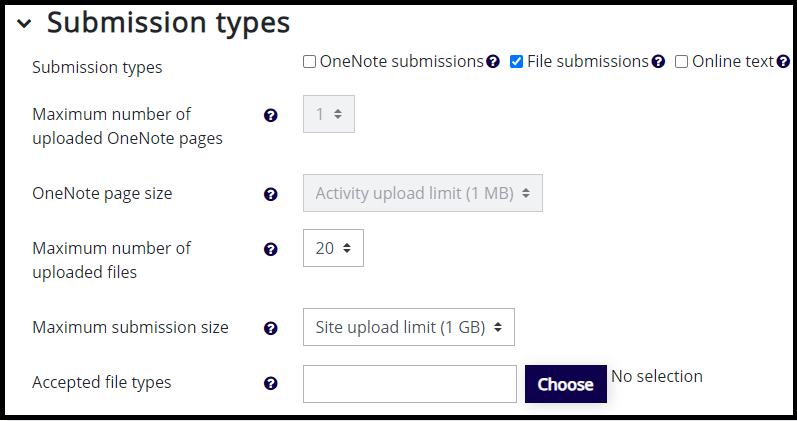
Creating An Assignment Activity Moodle
Comments are closed.