Structure Of Glucose And Fructose Molecular Formula Of Glucose And Fructose

Illustration Of Biology And Chemistry Fructose Molecular Molecular We explored structure of glucose and fructose —their molecular formula, open chain and ring forms, differences, and real life uses. understanding these structures helps you in topics like metabolism, food chemistry, and practical testing. Both fructose and glucose are simple sugars of monosaccharides. when digested, both starch and sugar, whether sucrose or high fructose corn syrup (hcfs), produce big quantities of glucose.
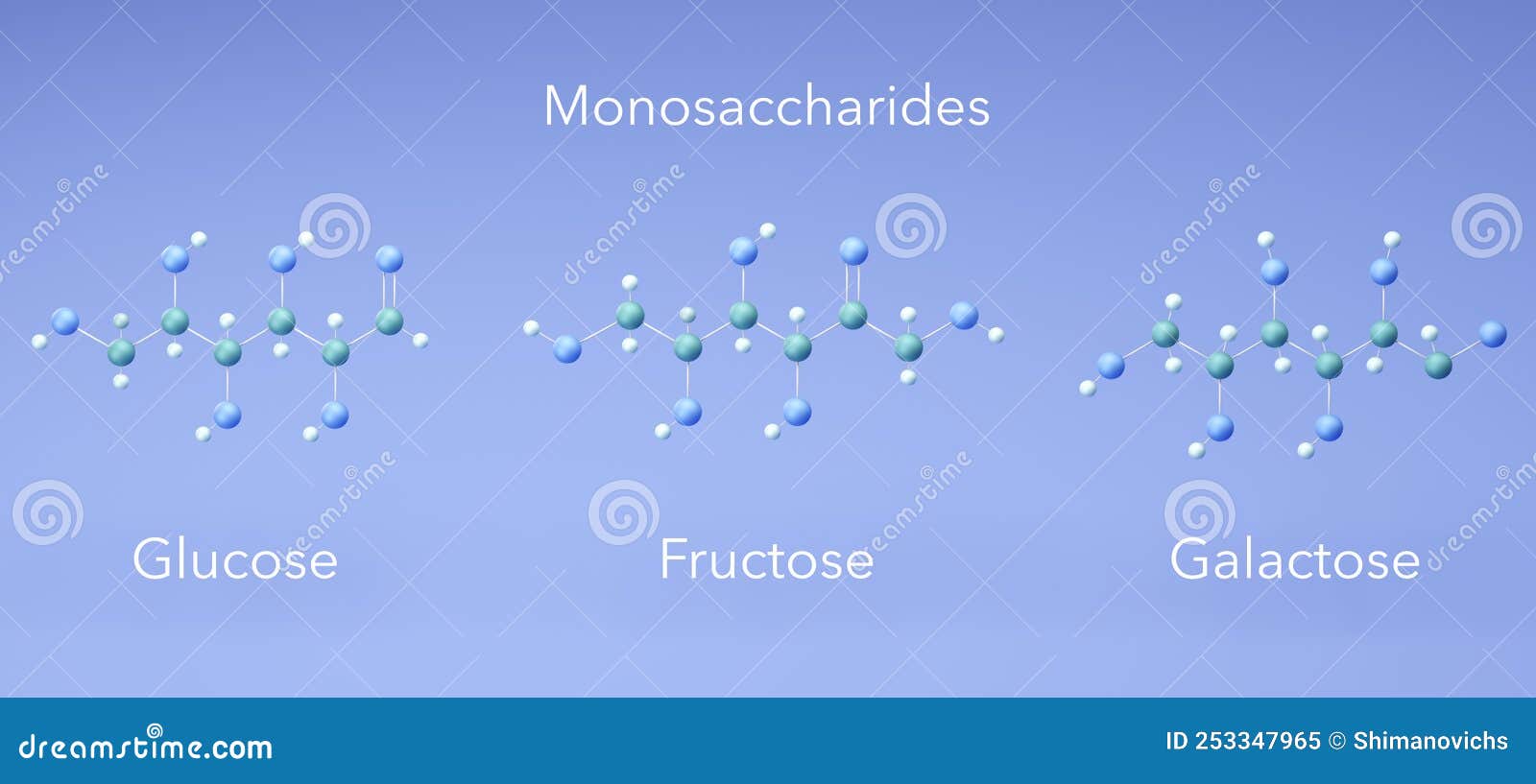
Monosaccharide Glucose Fructose Galactose Molecular Structures 3d The chemical or structural formula of fructose is as same as that of glucose, i.e., c 6 h 12 o 6. but they are stereogenically different from each other hence, isomers to each other. Learn about monosaccharides, glucose and fructose. study fischer & haworth structure, preparation, properties, reactions and differences. Glucose is a simple sugar (monosaccharide) generated during phosynthesis involving water, carbon and sunlight in plants. it is produced in humans via hepatic gluconeogenesis and breakdown of polymeric glucose forms (glycogenolysis). Galactose is a monosaccharide and has the same chemical formula as glucose, i.e., c6h12o6. it is similar to glucose in its structure, differing only in the position of one hydroxyl group.

Sucrose Fructose And Glucose Molecular Chemical Structural Formula Glucose is a simple sugar (monosaccharide) generated during phosynthesis involving water, carbon and sunlight in plants. it is produced in humans via hepatic gluconeogenesis and breakdown of polymeric glucose forms (glycogenolysis). Galactose is a monosaccharide and has the same chemical formula as glucose, i.e., c6h12o6. it is similar to glucose in its structure, differing only in the position of one hydroxyl group. Glucose and fructose are both sugars that share the same molecular formula, c₆h₁₂o₆, but they are not exactly the same in structure. even though they contain the same number and type of atoms, their functional groups are different. Both glucose and fructose are monosaccharides and structural isomers, sharing the chemical formula c ₆h ₁₂o ₆ but differing in atomic arrangement. in aqueous solutions, glucose predominantly forms a six membered pyranose ring, while fructose typically forms a five membered furanose ring. Answer: because they have the same molecular formula, c 6 h 12 o 6, but distinct functional groups in their chemical formula, glucose and fructose are functional isomers of each other. These sugars are the structural isomers of one another and have only one difference that glucose contains aldehyde functional group and fructose contains ketone functional group.
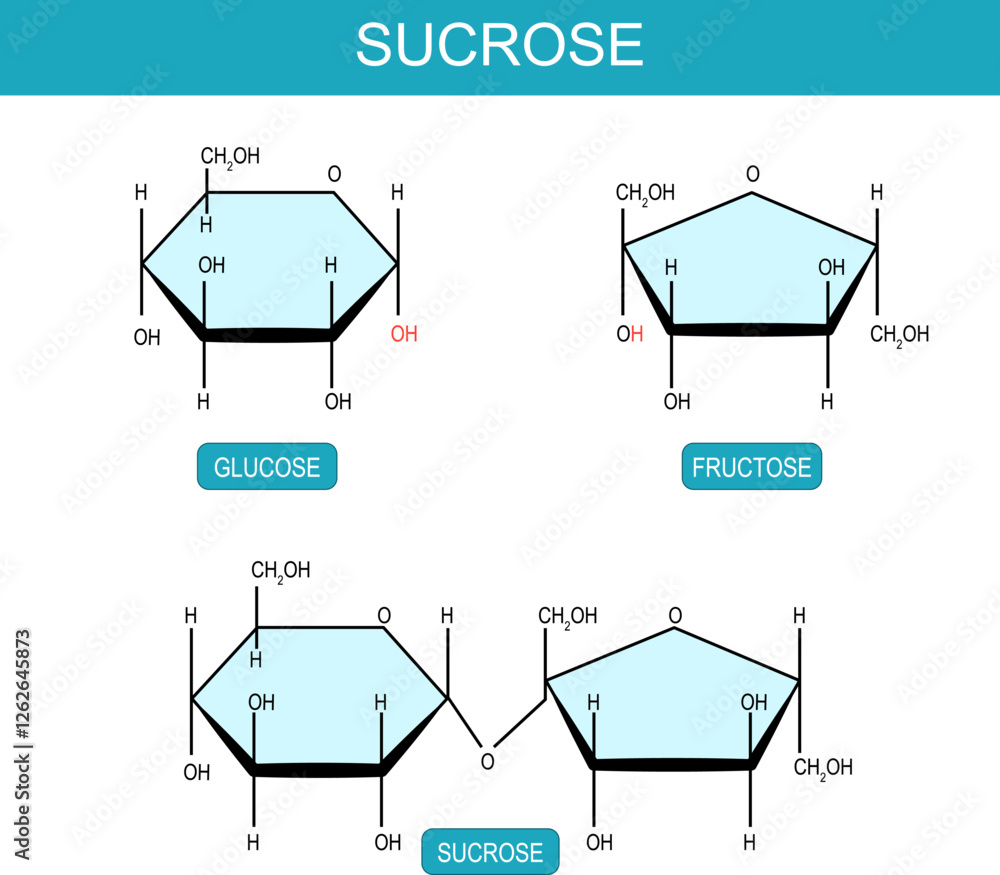
Sucrose Fructose And Glucose Molecular Chemical Structural Formula Glucose and fructose are both sugars that share the same molecular formula, c₆h₁₂o₆, but they are not exactly the same in structure. even though they contain the same number and type of atoms, their functional groups are different. Both glucose and fructose are monosaccharides and structural isomers, sharing the chemical formula c ₆h ₁₂o ₆ but differing in atomic arrangement. in aqueous solutions, glucose predominantly forms a six membered pyranose ring, while fructose typically forms a five membered furanose ring. Answer: because they have the same molecular formula, c 6 h 12 o 6, but distinct functional groups in their chemical formula, glucose and fructose are functional isomers of each other. These sugars are the structural isomers of one another and have only one difference that glucose contains aldehyde functional group and fructose contains ketone functional group.
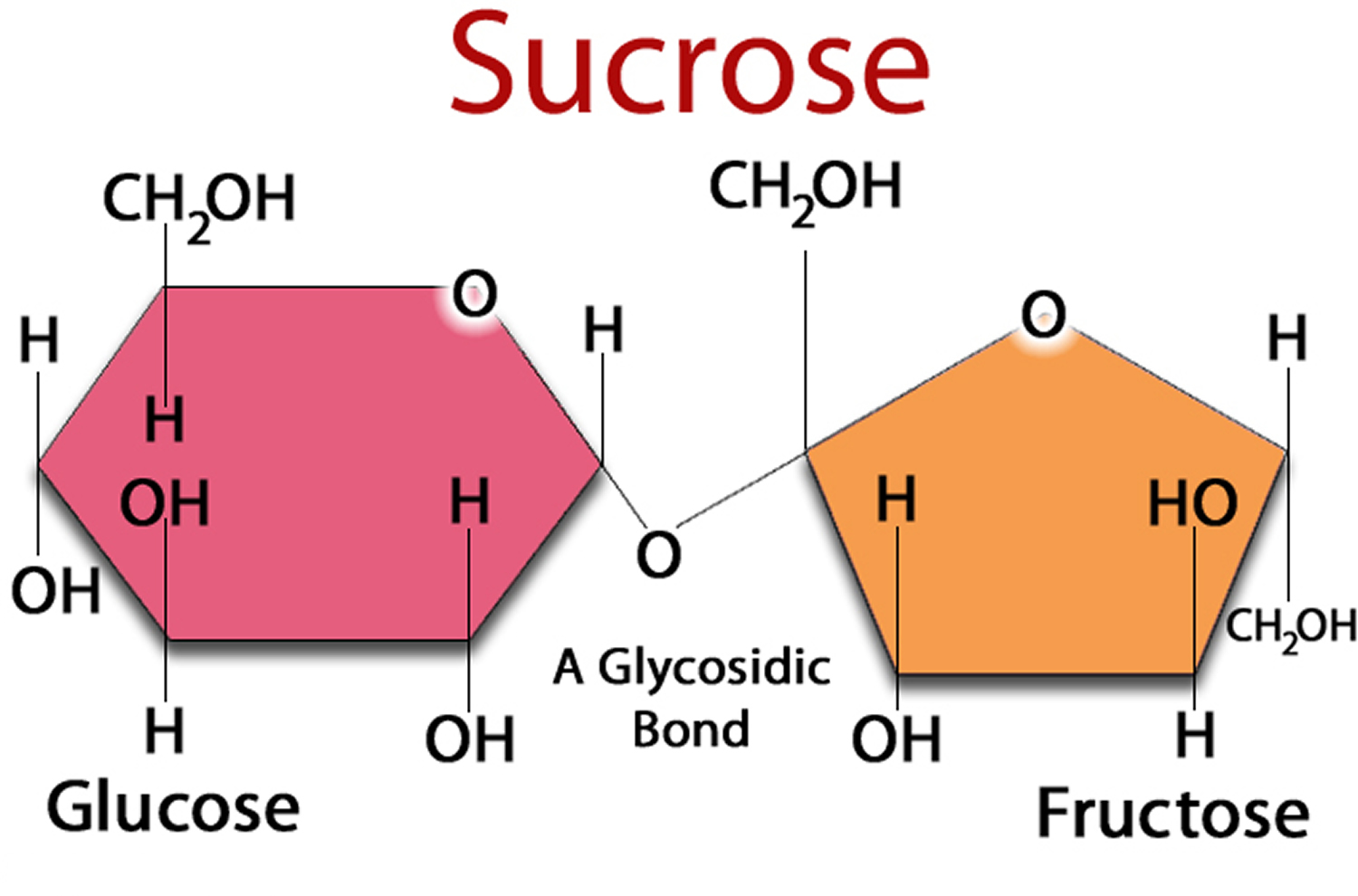
Fructose Molecular Structure Answer: because they have the same molecular formula, c 6 h 12 o 6, but distinct functional groups in their chemical formula, glucose and fructose are functional isomers of each other. These sugars are the structural isomers of one another and have only one difference that glucose contains aldehyde functional group and fructose contains ketone functional group.
Comments are closed.