Structure Of Diamond%f0%9f%92%8e Made By Me So Hard %f0%9f%a5%b5 Viralvideos Trending Study Learning Shorts

Happy Diwali Marathi Love Viralvideos Trending Chanchalnath Youtube Diamond is a mineral form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. diamond is a tasteless, odorless, strong, brittle solid, a poor conductor of electricity, colorless in pure form, and insoluble in water. The diamond cubic crystal structure is based on the face centered cubic bravais lattice (you can imagine it as two fcc unit cells, offset by ¼). there are 8 atoms per unit cell, and each atom is tetrahedrally coordinated so that it has 4 nearest neighbors.

Just Experiments Day 36 Of Learning Science Pradeep Shekhawat The so called diamond like crystal structure has received a great deal of attention because of its large calculated width of the band gap, and the property that the photonic band gap already opens at an index of refraction contrast of 1.9 compared to 2.8 for fcc air sphere crystals. Diamond has a cubic crystal structure where each carbon atom is bonded to four other carbon atoms in a tetrahedral arrangement. this structure consists of two interpenetrating face centered cubic lattices that are displaced relative to each other. Diamond is a naturally occurring mineral made of carbon atoms organized in a crystalline lattice structure. as a result, it has a number of distinct physical characteristics. First, we summarize the atomic structures as well as the synthesis methods of diamond. we then also discuss novel properties and potential applications based on these properties of diamond. finally, the outlook for diamond is also presented.
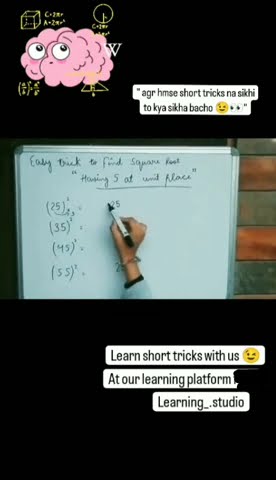
Drne Ki Kya Hai Baat Ab Team Learning Studio Hai Aapke Sath ёяшк Viral Diamond is a naturally occurring mineral made of carbon atoms organized in a crystalline lattice structure. as a result, it has a number of distinct physical characteristics. First, we summarize the atomic structures as well as the synthesis methods of diamond. we then also discuss novel properties and potential applications based on these properties of diamond. finally, the outlook for diamond is also presented. The structural unit of diamond consists of eight atoms, fundamentally arranged in a cube. this network is very stable and rigid, which is why diamonds are so very hard and have a high melting point. virtually all carbon on earth comes from the stars. Here we provide a comprehensive study of the n type sctd of the diamond by using first principles calculations based on the density functional theory. In diamond, each carbon shares electrons with four other carbon atoms forming four single bonds. in the diagram some carbon atoms only seem to be forming two bonds (or even one bond), but that's not really the case. we are only showing a small bit of the whole structure. The crystal structure of diamond is equivalent to a face centred cubic (fcc) lattice, with a basis of two identical carbon atoms: one at (0, 0, 0) and the other at (1 4, 1 4, 1 4), where the coordinates are given as fractions along the cube sides.
Comments are closed.