Statistical Analysis In Clinical Trials Epitech Research

3 Statistical Approaches To Analysis Of Small Clinical Trials Small This article aims to explain the key aspects of statistical analysis in clinical trials, offering a clear and detailed guide to various methodologies such as sequential analysis, hierarchical models, and bayesian methods. This review explores the critical role of statistical analysis in interpreting randomized controlled trials (rcts), focusing on how these methods are used to evaluate the efficacy and safety of clinical interventions.

A Comprehensive Guide To Analyzing Clinical Trial Data Pdf The analysis of clinical trial data is vital for determining the true effects of treatments and differentiating these effects from random variation. two key statistical methodologies are discussed: descriptive and inferential. This document provides guidance on the design, conduct, analysis and evaluation of clinical trials of an investigational product in the context of its overall clinical development. The proliferation of statistical research in the area of clinical trials coupled with the critical role of clinical research in the drug approval process and health care in general. This reporting guideline recommends a minimum set of 55 items that should be addressed and included in statistical analysis plans for clinical trials.

Clinical Research Epitech The proliferation of statistical research in the area of clinical trials coupled with the critical role of clinical research in the drug approval process and health care in general. This reporting guideline recommends a minimum set of 55 items that should be addressed and included in statistical analysis plans for clinical trials. Here, three experts discuss the intricacies of clinical trial statistical planning and analysis as well as common issues that arise and emerging trends. Additionally, much of the literature on car t cell therapy outcomes is focused on clinical trials which differ from real world settings in long term follow up of these patients. there are some statistical challenges in analyzing car t cell therapy data, such as defining patients' response to therapy and quantifying the duration of responses. “this book provides an excellent description of the methods and applications of statistics to design clinical trials and to understand and evaluate data at different stages of clinical trials. Epitech completed pre clinical trials in 2017 and published the results in 2020 which demonstrated that rms treatment decreases epithelial corneal erosions in a rabbit model of exposure keratopathy, with no indication of pathological changes.
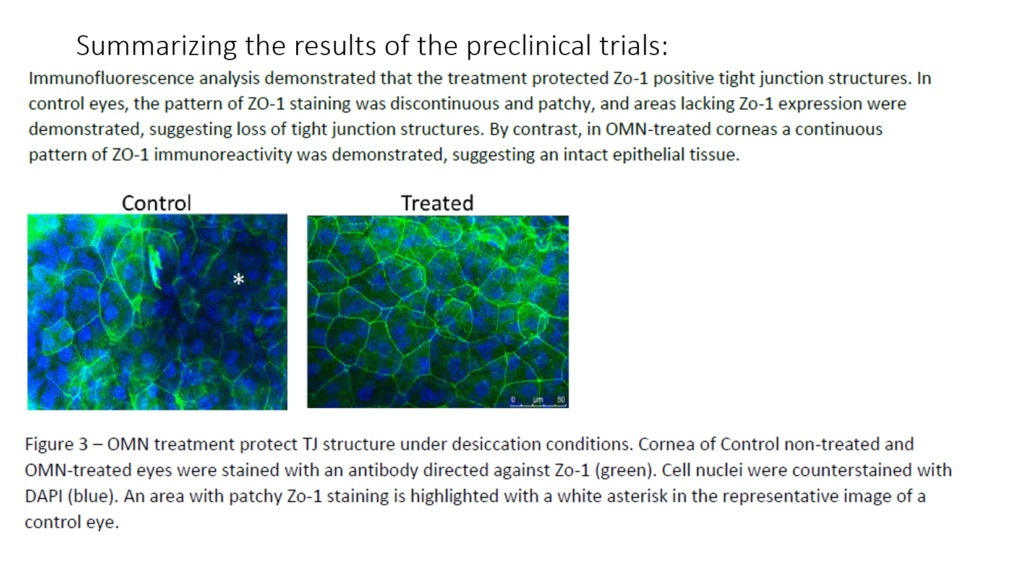
Clinical Research Epitech Here, three experts discuss the intricacies of clinical trial statistical planning and analysis as well as common issues that arise and emerging trends. Additionally, much of the literature on car t cell therapy outcomes is focused on clinical trials which differ from real world settings in long term follow up of these patients. there are some statistical challenges in analyzing car t cell therapy data, such as defining patients' response to therapy and quantifying the duration of responses. “this book provides an excellent description of the methods and applications of statistics to design clinical trials and to understand and evaluate data at different stages of clinical trials. Epitech completed pre clinical trials in 2017 and published the results in 2020 which demonstrated that rms treatment decreases epithelial corneal erosions in a rabbit model of exposure keratopathy, with no indication of pathological changes.

Statistical Analysis For Clinical Trials Medical Research Freelance “this book provides an excellent description of the methods and applications of statistics to design clinical trials and to understand and evaluate data at different stages of clinical trials. Epitech completed pre clinical trials in 2017 and published the results in 2020 which demonstrated that rms treatment decreases epithelial corneal erosions in a rabbit model of exposure keratopathy, with no indication of pathological changes.
Comments are closed.