States Of Matter Liquid Examples

States Of Matter Diagram Solid Liquid And Gas Get 10 examples of solids, liquids, gases, and plasma. these are the four main states or phases of matter. learn about phase transitions. What is a liquid. what are its facts and characteristics. how are the particles arranged. check out its types with examples and a diagram.
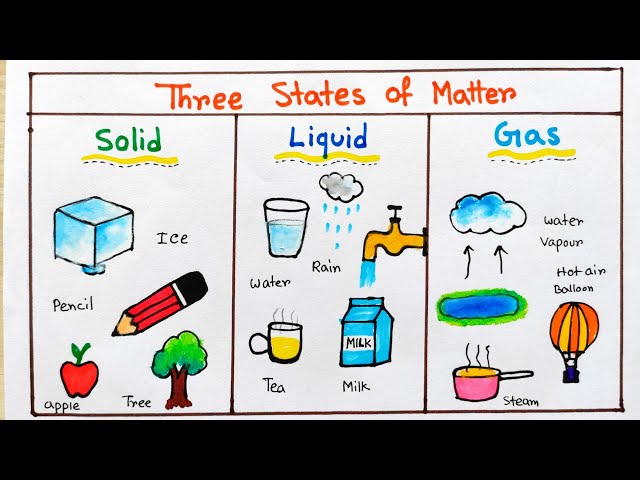
States Of Matter Liquid Examples States Of Matter 7g Teaching Liquids may be divided into two general categories: pure liquids and liquid mixtures. on earth, water is the most abundant liquid, although much of the water with which organisms come into contact is not in pure form but is a mixture in which various substances are dissolved. Common examples of the liquid state include water, mercury, alcohol, oil, and bromine. these substances demonstrate the essential characteristics of liquids as described by their particle arrangement and dynamic behavior. Liquids can be categorized into pure liquids and liquid mixtures, with water being a prevalent example. liquid mixtures, such as blood and seawater, involve dissolved substances, showcasing the diversity of compositions in liquid states. Most known matter in the universe is either gaseous (as interstellar clouds) or plasma (as stars). only two elements are liquid at standard conditions for temperature and pressure: mercury and bromine. four more elements have melting points slightly above room temperature: francium, caesium, gallium and rubidium. [1].
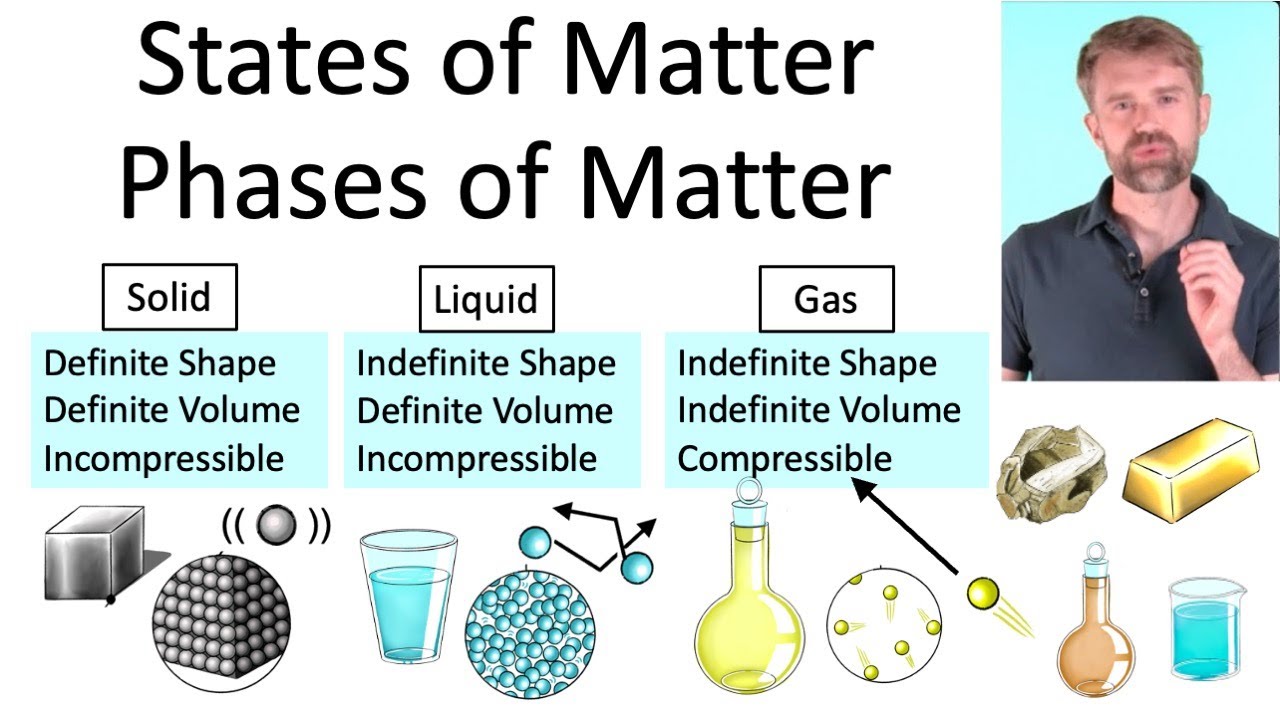
States Of Matter Liquid Examples States Of Matter 7g Teaching Liquids can be categorized into pure liquids and liquid mixtures, with water being a prevalent example. liquid mixtures, such as blood and seawater, involve dissolved substances, showcasing the diversity of compositions in liquid states. Most known matter in the universe is either gaseous (as interstellar clouds) or plasma (as stars). only two elements are liquid at standard conditions for temperature and pressure: mercury and bromine. four more elements have melting points slightly above room temperature: francium, caesium, gallium and rubidium. [1]. The state a given substance exhibits is also a physical property. some substances exist as gases at room temperature (oxygen and carbon dioxide), while others, like water and mercury metal, exist as liquids. Discover how solids, liquids and gases differ in chemistry. learn how particle behavior defines each state and see real‑world examples. Three states of matter exist: solid, liquid, and gas. solids have a definite shape and volume. liquids have a definite volume, but take the shape of the container. gases have no definite shape or …. Examples of liquids include water, oil, and blood. a liquid is a state of matter that has a definite volume, but no fixed shape. in other words, a liquid takes the shape of its container. liquids consist of atoms or molecules that are loosely connected by intermolecular bonds.
Comments are closed.